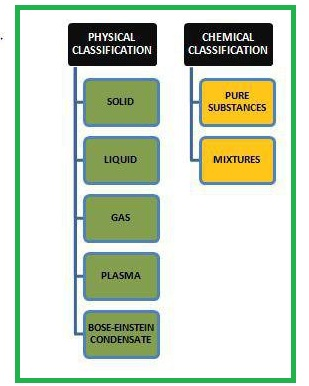
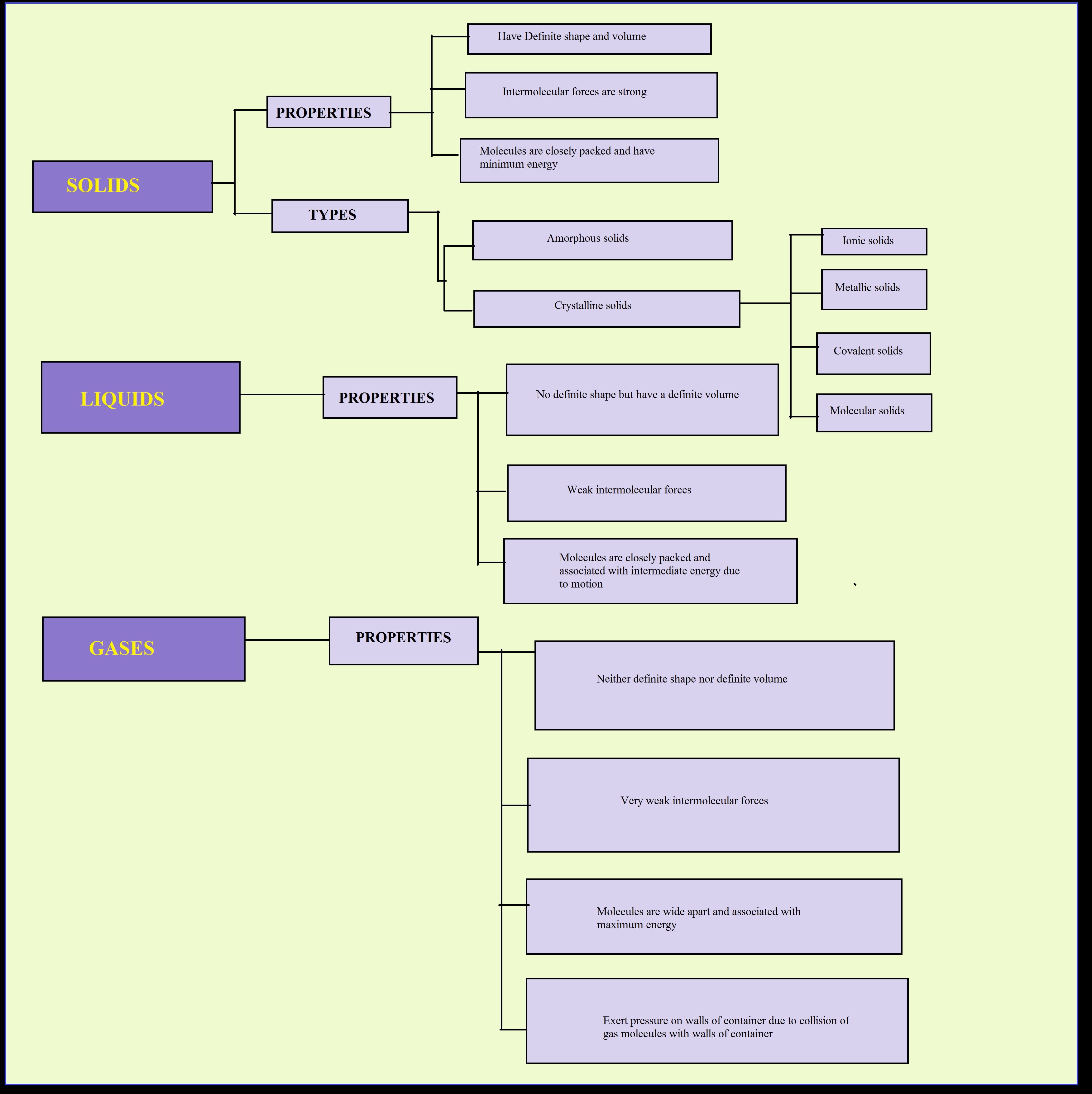
Physical Changes :
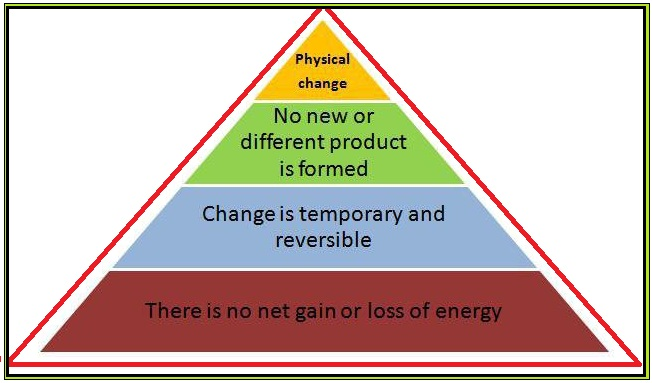
A physical change is a change in which the composition of a substance is not affected, i.e. no new substance is formed. The physical changes are reversible.
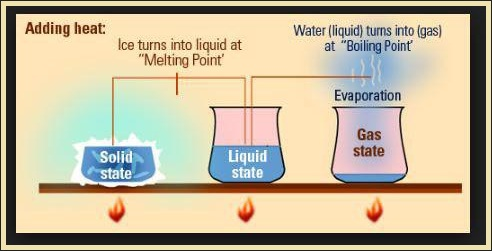
Examples : (i) Interconversion of states is a physical change because these changes occur without a change in composition and no change in the chemical nature of the substance.
(ii) When an ordinary piece of iron is magnetised in the presence of a strong magnetic field. Since colour, density, mass and the composition remains the same. Therefore, magnetisation is a physical change. Magnetisation can be reversed by heating the piece of iron.
(iii) Evaporation, distillation, sublimation, condensation and crystallisation are examples of physical changes.
(iv) The blue colour of copper sulphate crystal disappears when it is heated strongly because on heating water of crystallisation of crystal is lost. It regains its blue colour when moistened with a drop of water.
`undersettext(Blue) (CuSO_4*5H_2O) oversettext(Heat)→ undersettext(white) (CuSO_4) +5H_2O`
It is an example of a physical change.
Some facts about the physical changes are
(i) Physical changes are temporary.
(ii) Composition of the constituent of matter remains same during physical change i.e. constituent particles of matter does not change.
(iii) Physical changes are due to the change in physical properties like density, volume, state temporarily.
`text(Note)`
► Amount of space that a substance or object occupies is called volume of that substance.
► Density is the degree of compactness of a substance. It is defined as mass per unit volume.
i.e. `text(Density) = text(mass)/text(volume)`
Examples : (i) Interconversion of states is a physical change because these changes occur without a change in composition and no change in the chemical nature of the substance.
(ii) When an ordinary piece of iron is magnetised in the presence of a strong magnetic field. Since colour, density, mass and the composition remains the same. Therefore, magnetisation is a physical change. Magnetisation can be reversed by heating the piece of iron.
(iii) Evaporation, distillation, sublimation, condensation and crystallisation are examples of physical changes.
(iv) The blue colour of copper sulphate crystal disappears when it is heated strongly because on heating water of crystallisation of crystal is lost. It regains its blue colour when moistened with a drop of water.
`undersettext(Blue) (CuSO_4*5H_2O) oversettext(Heat)→ undersettext(white) (CuSO_4) +5H_2O`
It is an example of a physical change.
Some facts about the physical changes are
(i) Physical changes are temporary.
(ii) Composition of the constituent of matter remains same during physical change i.e. constituent particles of matter does not change.
(iii) Physical changes are due to the change in physical properties like density, volume, state temporarily.
`text(Note)`
► Amount of space that a substance or object occupies is called volume of that substance.
► Density is the degree of compactness of a substance. It is defined as mass per unit volume.
i.e. `text(Density) = text(mass)/text(volume)`