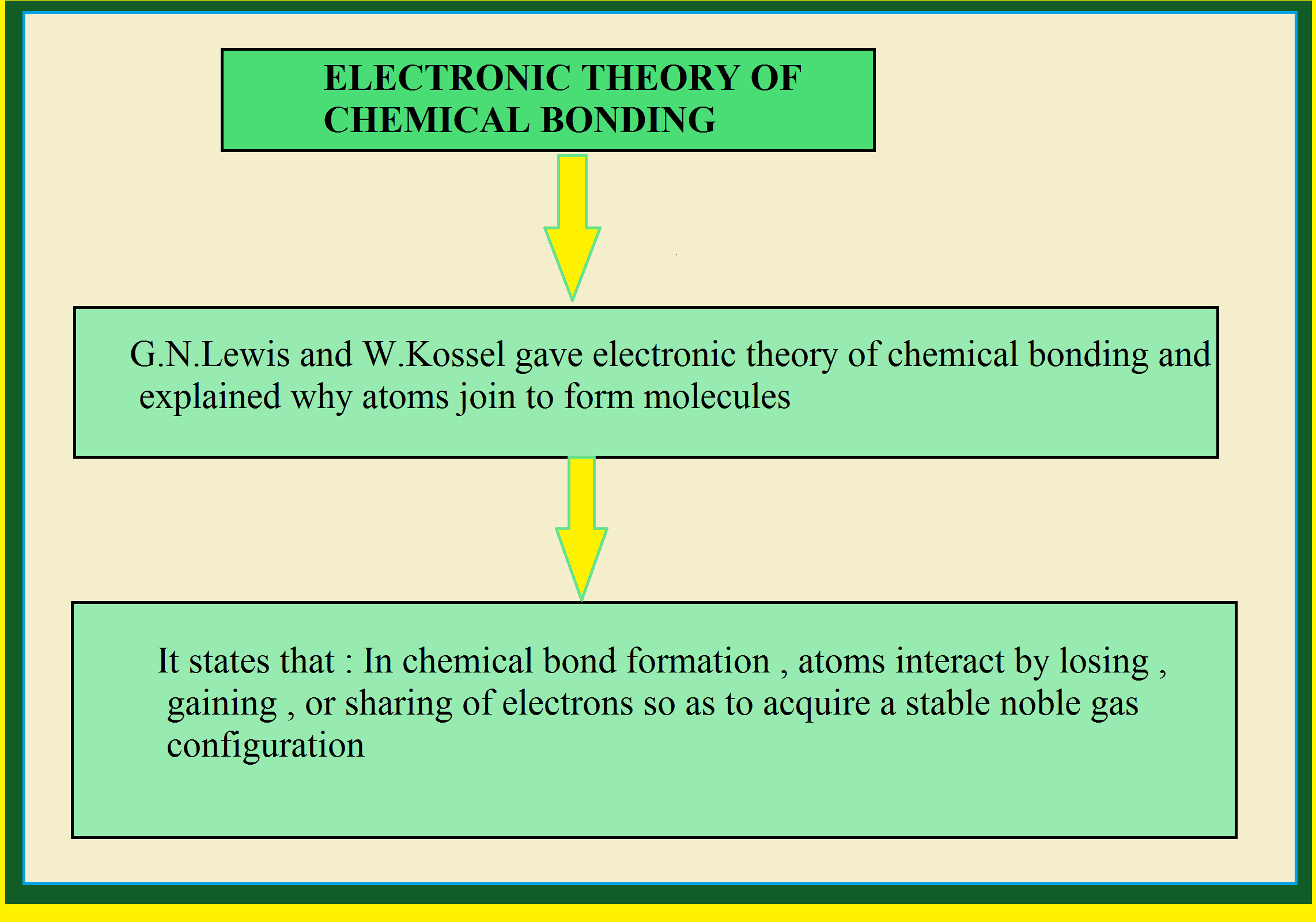
`=>` The capacity of an element to form chemical bonds is known as its valency. The valency of an element is the combining capacity of an element.
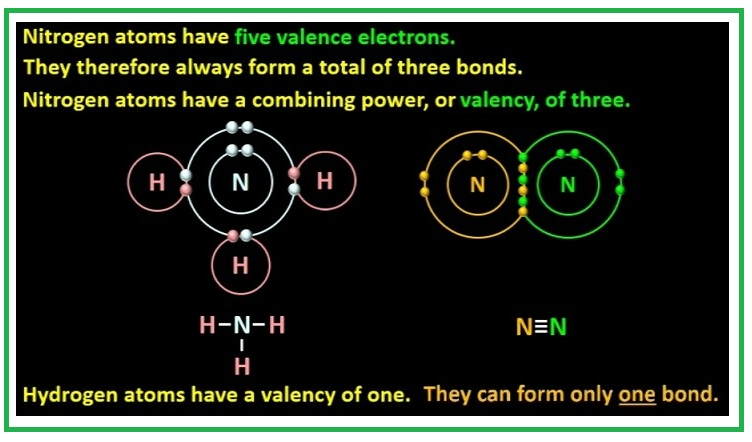
`=>` The valency of an element decides the number of other atoms which can combine with one atom of that element. e.g. the valency of carbon is 4 and that of hydrogen is 1. So, one atom of carbon can combine with four atoms of hydrogen to form a methane molecule `CH_4` .
`=>` Valency of metal = Number of valence electrons in its atom. e.g. sodium (Na) = 2, 8, 1
`:.` Valency of sodium = 1
`=>` Valency of non-metal = 8- Number of valence electrons
in its atom. e.g. chlorine `Cl_17` = 2, 8, 7
`:. ` Valency of` Cl = 8-7 =1`
`=>` Valency of non-metal = 8- Number of valence electrons in its atom. e.g. chlorine `Cl_17 = 2, 8, 7`
`:.` Valency of `Cl = 8-7 =1`
Valencies of some important elements are as follows :
(i) H, Cl, Br, I, Cu (ous) etc. are monovalent as their valency is 1 .
(ii) O, S, Mg, Ca Zn, Fe (ous), Cu (ic), Pb, Co, Ni etc.
are divalent as their valency is 2.
(iii) N, P, Al, Cr, B etc are trivalent as their valency is 3.
(iv) C, Si etc are tetravalent as their valency is 4.
Some elements e.g., Cu, Fe etc. exhibit more than one valencies (variable valency). Cu and Hg exhibit +1 and +2 valencies and Fe exhibits +2 and +3 valencies.
`=>` The capacity of an element to form chemical bonds is known as its valency. The valency of an element is the combining capacity of an element.
`=>` The valency of an element decides the number of other atoms which can combine with one atom of that element. e.g. the valency of carbon is 4 and that of hydrogen is 1. So, one atom of carbon can combine with four atoms of hydrogen to form a methane molecule `CH_4` .
`=>` Valency of metal = Number of valence electrons in its atom. e.g. sodium (Na) = 2, 8, 1
`:.` Valency of sodium = 1
`=>` Valency of non-metal = 8- Number of valence electrons
in its atom. e.g. chlorine `Cl_17` = 2, 8, 7
`:. ` Valency of` Cl = 8-7 =1`
`=>` Valency of non-metal = 8- Number of valence electrons in its atom. e.g. chlorine `Cl_17 = 2, 8, 7`
`:.` Valency of `Cl = 8-7 =1`
Valencies of some important elements are as follows :
(i) H, Cl, Br, I, Cu (ous) etc. are monovalent as their valency is 1 .
(ii) O, S, Mg, Ca Zn, Fe (ous), Cu (ic), Pb, Co, Ni etc.
are divalent as their valency is 2.
(iii) N, P, Al, Cr, B etc are trivalent as their valency is 3.
(iv) C, Si etc are tetravalent as their valency is 4.
Some elements e.g., Cu, Fe etc. exhibit more than one valencies (variable valency). Cu and Hg exhibit +1 and +2 valencies and Fe exhibits +2 and +3 valencies.