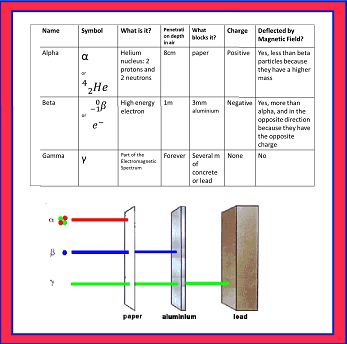
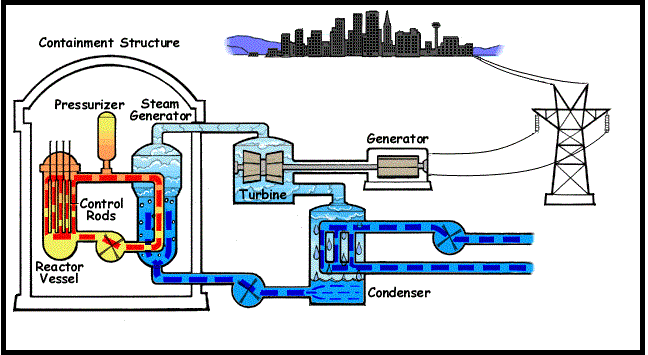
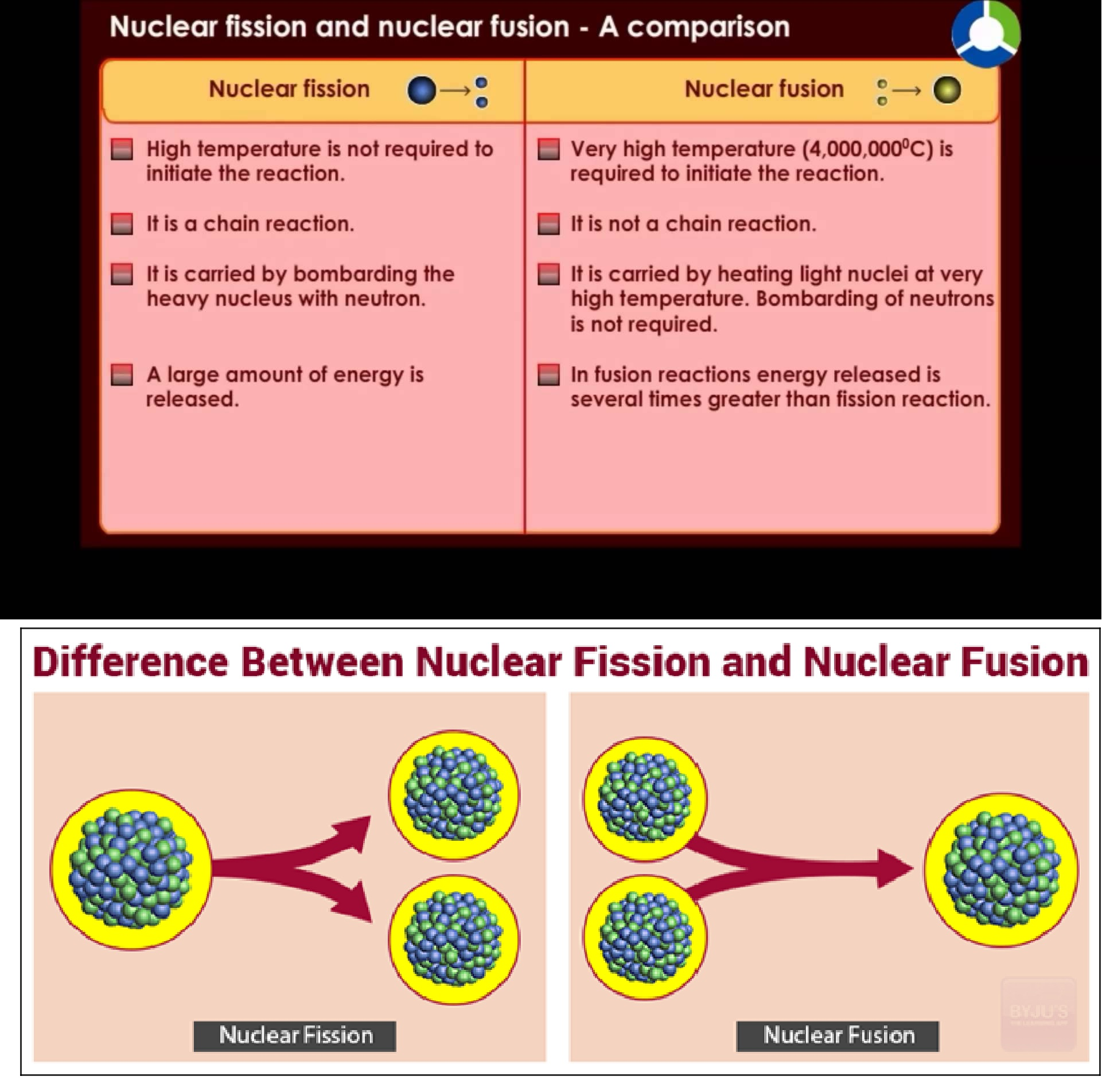
Nuclear Forces
Definition : – Nuclear force is one of the four fundamental forces of nature, the others being gravitational and electromagnetic forces. In fact, being 10 million times stronger than the chemical binding forces, they are also known as the strong forces. In this section, we will discuss this force in detail.
Nuclear Force
Nuclear force is the force that binds the neutrons and protons in a nucleus together. This force can exist between neutrons and neutrons, neutrons and protons or protons and protons. This force is what holds the nucleus together. The charge of protons, which is `+1e`, tends to push them away from each other with a strong electric field repulsive force, following Coulomb’s law. But nuclear force is strong enough to overcome that resistance at short range and keep them together.
Properties
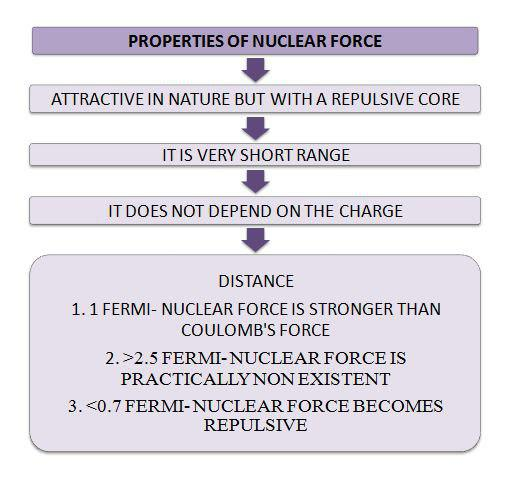
`=>` It is attractive in nature but with a repulsive core. That is the reason that the nucleus is held together without collapsing in itself.
`=>` The range of nuclear force is very short. At 1 Fermi, the distance between particles in a nucleus is extremely small. At this range, the nuclear force is much stronger than the repulsive Coulomb’s force that pushes the particles away. However, if the distance is anything more than 2.5 Fermi, nuclear force is practically non-existent.
`=>` Nuclear force is identical for all nucleons. It does not matter if it is a neutron or proton, once the Coulomb resistance is taken into consideration, nuclear force affects everything in the same way.
`=>` At a distance less than 0.7 Fermi, this force becomes repulsive. It is one of the most interesting properties of nuclear force, as this repulsive component of the force is what decides the size of the nucleus. The nucleons come closer to each other till the point that the force allows, after which they cannot come any closer because of the repulsive property of the force.
Nuclear Force
Nuclear force is the force that binds the neutrons and protons in a nucleus together. This force can exist between neutrons and neutrons, neutrons and protons or protons and protons. This force is what holds the nucleus together. The charge of protons, which is `+1e`, tends to push them away from each other with a strong electric field repulsive force, following Coulomb’s law. But nuclear force is strong enough to overcome that resistance at short range and keep them together.
Properties
`=>` It is attractive in nature but with a repulsive core. That is the reason that the nucleus is held together without collapsing in itself.
`=>` The range of nuclear force is very short. At 1 Fermi, the distance between particles in a nucleus is extremely small. At this range, the nuclear force is much stronger than the repulsive Coulomb’s force that pushes the particles away. However, if the distance is anything more than 2.5 Fermi, nuclear force is practically non-existent.
`=>` Nuclear force is identical for all nucleons. It does not matter if it is a neutron or proton, once the Coulomb resistance is taken into consideration, nuclear force affects everything in the same way.
`=>` At a distance less than 0.7 Fermi, this force becomes repulsive. It is one of the most interesting properties of nuclear force, as this repulsive component of the force is what decides the size of the nucleus. The nucleons come closer to each other till the point that the force allows, after which they cannot come any closer because of the repulsive property of the force.