Electrovalent or Ionic Bond
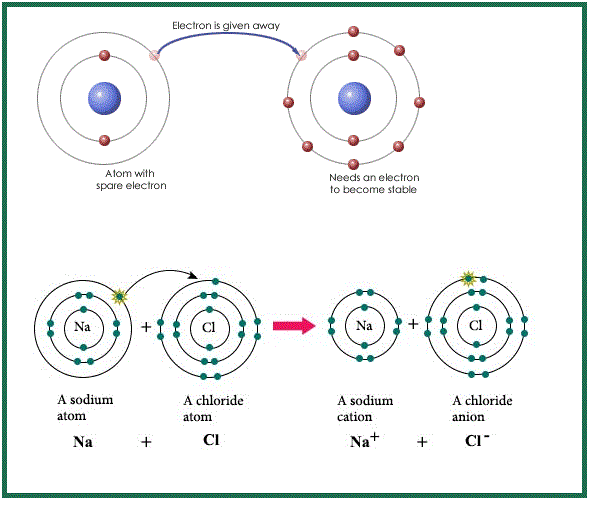
`=>` The chemical bond formed by transfer of electron from a metal to a non- metal is known as ionic or electrovalent bond.
`=>` It is formed between most electropositive elements i.e. metals (like, Na, K, Ca, Ba, Mg etc.) and most electronegative elements i.e. non-metals (like `O`, `F`, `N`, `S`, `Cl`, `Br` etc.).
`=>` The number of electrons lost or gained by the element is called electrovalency of the element which is equal to charge of cation or anion respectively and these types of compounds are called electrovalent compounds.
`=>` Cations are formed by losing electrons by electropositive elements and anions are formed by taking up electrons by electronegative elements.
`=>` Bond is generally ionic, if the difference in electronegativity of the atoms is around `1.9` and more.
`=>` It is formed between most electropositive elements i.e. metals (like, Na, K, Ca, Ba, Mg etc.) and most electronegative elements i.e. non-metals (like `O`, `F`, `N`, `S`, `Cl`, `Br` etc.).
`=>` The number of electrons lost or gained by the element is called electrovalency of the element which is equal to charge of cation or anion respectively and these types of compounds are called electrovalent compounds.
`=>` Cations are formed by losing electrons by electropositive elements and anions are formed by taking up electrons by electronegative elements.
`=>` Bond is generally ionic, if the difference in electronegativity of the atoms is around `1.9` and more.