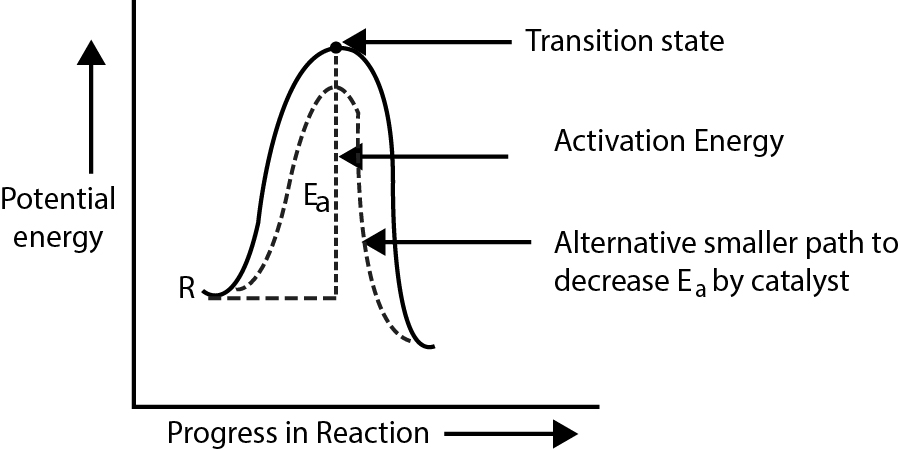
(i) Positive catalysis : The catalyst increases the rate of a reaction.
Examples: `2SO_2+O_2 overset(V_2O_5)-> 2SO_3` (Contact Process)
`C_2H_4 + H_2 underset(Delta) overset(N i) rightarrow C_2H_6` (Ethane)
(ii) Negative catalysis: (Inhibitor or retarder) : Chemical reactions are sometimes retarded by the presence of a foreign substance. The substance is known as a negative catalyst.
Examples: Alcohol, Acetanilide : Prevents oxidation of `Na_2SO_3` by air
`H_3PO_4` : Prevents decomposition of `H_2O_2`
(iii) Auto catalysis: In this type of catalysis, one of the product of the reaction catalyses the reaction. In the oxidation of oxalic acid by `KMnO_4`, `Mn^(2+)` ion formed is known to accelerate the reaction. So, when `KMnO_4` solution is run into warm solution of oxalic
acid (+ dil. ` H_2SO_4`), initially there is a time lag before decolourisation occurs; as more `KMnO_4` is added, the decolourisation becomes almost instantaneous.
(iv) Homogeneous catalysis: A catalytic process in which the catalyst is in the same phase as the reactant is called homogeneous catalysis.
`2SO_2(g)+O_2(g) overset(NO) -> 2SO_3(g)` (Lead chamber process)
`C_12H_22O_11(aq)+H_2O overset(H^+ (aq))-> C_6H_12O_6(aq)+C_6H_12O_6(aq)` (Inversion of cane sugar)
(v) Heterogeneous catalysis: A catalytic process in which the catalyst and the reactants are in different phases is called heterogeneous catalysis. This process is also called contact or surface catalysis.
Examples: (a) `2H_2O_2( l) overset(Pt)-> 2H_2O(l) + O_2(g) `
(b) `2SO_2(g) + O_2(g) overset( Pt quad text(asbestos)) rightarrow 2SO_3(g)`
(c) `N_2(g) + 3H_2(g) overset( Fe-Mo) rightarrow 2NH_3(g)`
(d) `4NH_3(g) + 5O_2(g) overset(Pt quad text(gauze)) rightarrow 4NO(g)+ 6H_2O(l)`
(e) `CO(g) + 2H_2(g) overset(ZnO + Cr_2O_3) rightarrow CH_3OH(l)`
(vi) Induced catalyst: When one reaction influences the rate of other reaction, which does not occur under ordinary conditions, the phenomenon is known as induced catalysis.
Examples of induced catalysis:
(a) Sodium arsenite solution is not oxidised by air. If, however, air is passed through a mixture of the solution of sodium arsenite and sodium sulphite, both of them undergo simultaneous oxidation. The oxidation of sodium sulphite, thus, induces the oxidation of sodium arsenite.
(b) The reduction of mercuric chloride (`HgCl_2`) with oxalic acid is very slow, but potassium permanganate is reduced readily with oxalic acid. If, however, oxalic acid is added to a mixture of `KMnO_4` and `HgCl_2`, both are reduced simultaneously. The reduction of potassium permanganate, thus, induces the reduction of `HgCl_2`.
(i) Positive catalysis : The catalyst increases the rate of a reaction.
Examples: `2SO_2+O_2 overset(V_2O_5)-> 2SO_3` (Contact Process)
`C_2H_4 + H_2 underset(Delta) overset(N i) rightarrow C_2H_6` (Ethane)
(ii) Negative catalysis: (Inhibitor or retarder) : Chemical reactions are sometimes retarded by the presence of a foreign substance. The substance is known as a negative catalyst.
Examples: Alcohol, Acetanilide : Prevents oxidation of `Na_2SO_3` by air
`H_3PO_4` : Prevents decomposition of `H_2O_2`
(iii) Auto catalysis: In this type of catalysis, one of the product of the reaction catalyses the reaction. In the oxidation of oxalic acid by `KMnO_4`, `Mn^(2+)` ion formed is known to accelerate the reaction. So, when `KMnO_4` solution is run into warm solution of oxalic
acid (+ dil. ` H_2SO_4`), initially there is a time lag before decolourisation occurs; as more `KMnO_4` is added, the decolourisation becomes almost instantaneous.
(iv) Homogeneous catalysis: A catalytic process in which the catalyst is in the same phase as the reactant is called homogeneous catalysis.
`2SO_2(g)+O_2(g) overset(NO) -> 2SO_3(g)` (Lead chamber process)
`C_12H_22O_11(aq)+H_2O overset(H^+ (aq))-> C_6H_12O_6(aq)+C_6H_12O_6(aq)` (Inversion of cane sugar)
(v) Heterogeneous catalysis: A catalytic process in which the catalyst and the reactants are in different phases is called heterogeneous catalysis. This process is also called contact or surface catalysis.
Examples: (a) `2H_2O_2( l) overset(Pt)-> 2H_2O(l) + O_2(g) `
(b) `2SO_2(g) + O_2(g) overset( Pt quad text(asbestos)) rightarrow 2SO_3(g)`
(c) `N_2(g) + 3H_2(g) overset( Fe-Mo) rightarrow 2NH_3(g)`
(d) `4NH_3(g) + 5O_2(g) overset(Pt quad text(gauze)) rightarrow 4NO(g)+ 6H_2O(l)`
(e) `CO(g) + 2H_2(g) overset(ZnO + Cr_2O_3) rightarrow CH_3OH(l)`
(vi) Induced catalyst: When one reaction influences the rate of other reaction, which does not occur under ordinary conditions, the phenomenon is known as induced catalysis.
Examples of induced catalysis:
(a) Sodium arsenite solution is not oxidised by air. If, however, air is passed through a mixture of the solution of sodium arsenite and sodium sulphite, both of them undergo simultaneous oxidation. The oxidation of sodium sulphite, thus, induces the oxidation of sodium arsenite.
(b) The reduction of mercuric chloride (`HgCl_2`) with oxalic acid is very slow, but potassium permanganate is reduced readily with oxalic acid. If, however, oxalic acid is added to a mixture of `KMnO_4` and `HgCl_2`, both are reduced simultaneously. The reduction of potassium permanganate, thus, induces the reduction of `HgCl_2`.