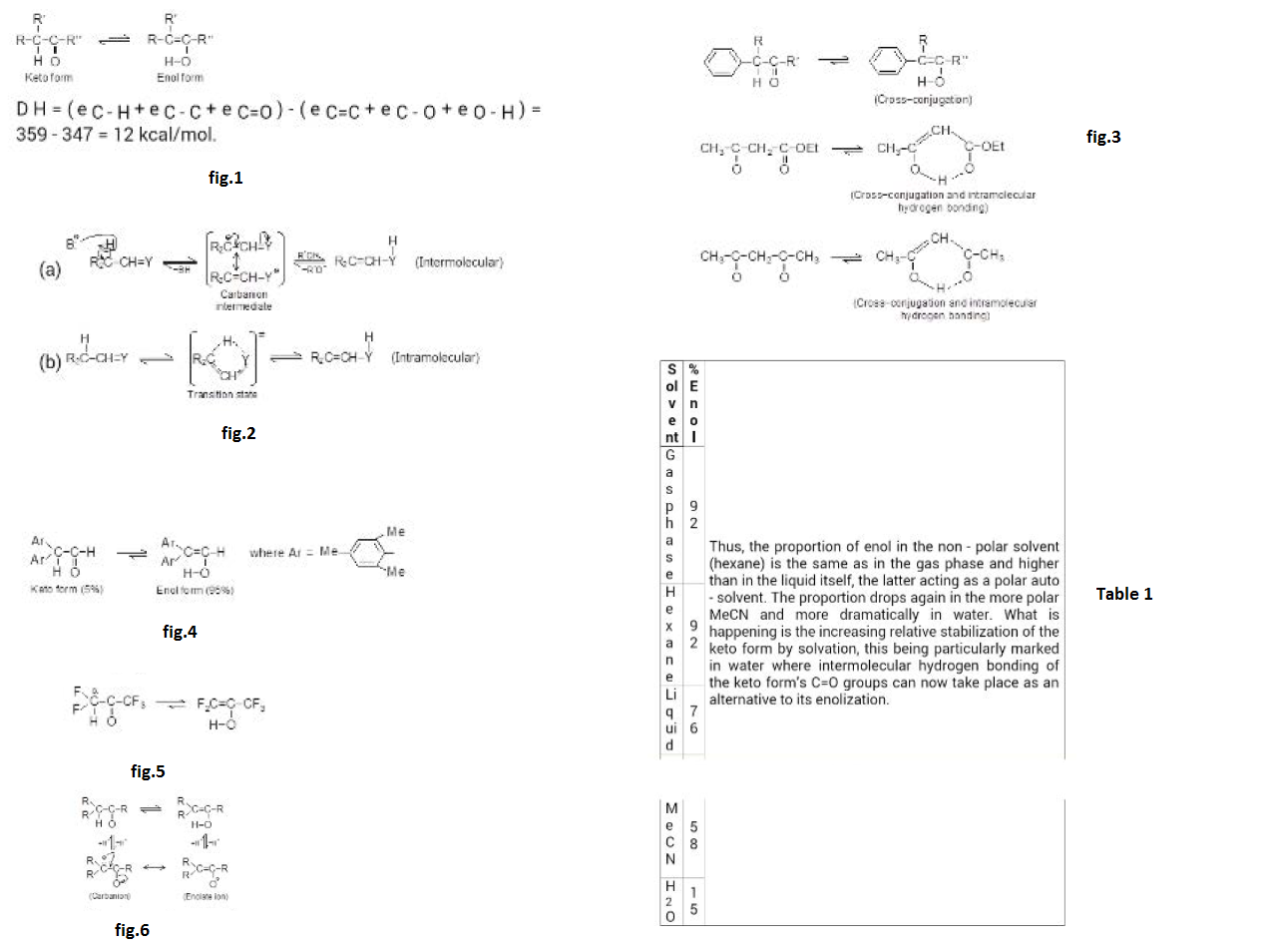
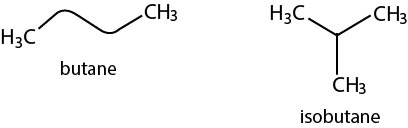Isomerism :
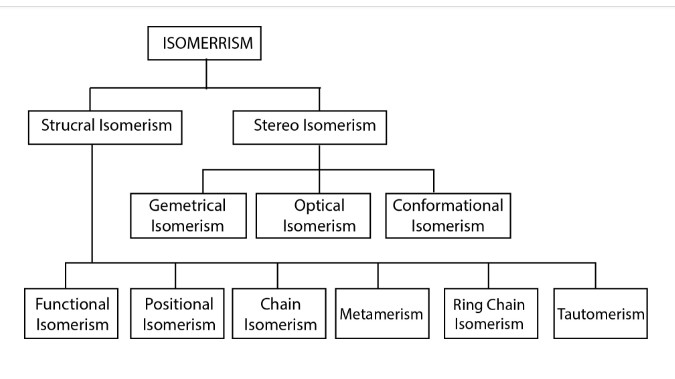
In the study of organic chemistry we come across many cases when two or more compounds are made of equal number of like atoms. A molecular formula does not tell the nature of organic compound; sometimes several organic compounds may have same molecular formula. The compounds possess the same molecular formula but differ from each other in physical or chemical properties, are called isomers and the phenomenon is termed isomerism (Greek, isos = equal; meres = parts). Since isomers have the same molecular formula, the difference in their properties must be due to different modes of the combination or arrangement of atoms within the molecule. Broadly speaking, isomerism is of two types.
i) Structural Isomerism ii) Stereoisomerism
i) Structural isomerism : When the isomerism is simply due to difference in the arrangement of atoms within the molecule without any reference to space, the phenomenon is termed structural isomerism. In other words, while they have same molecular formulas they possess different structural formula. This type of isomerism which arises from difference in the structure of molecules, includes:
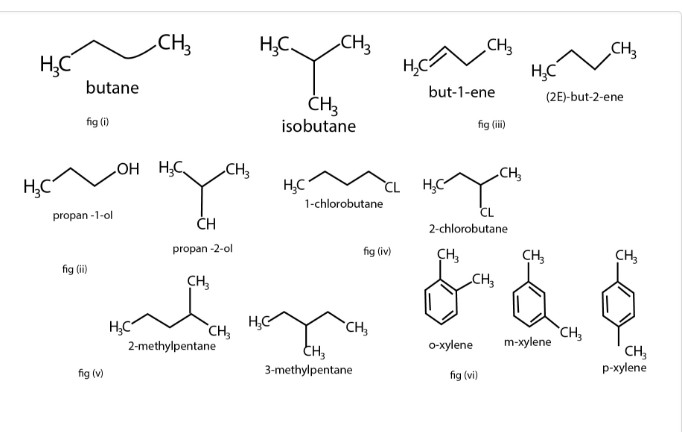
a) Chain or Nuclear Isomerism
b) Positional Isomerism
c) Metamerism
d) Functional Isomerism
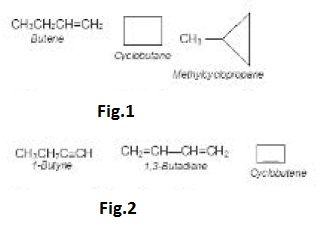
e) Ring Chain Isomerism
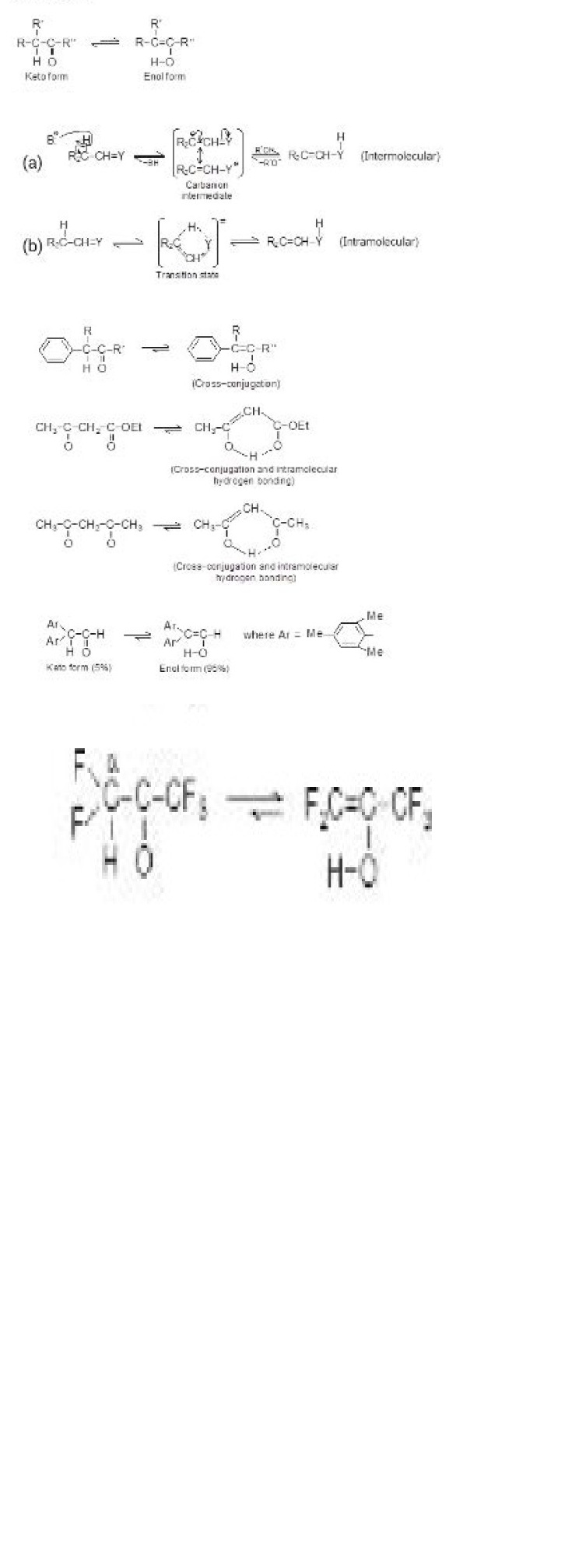
f) Tautomerism
ii) Stereoisomerism : When isomerism is caused by the different arrangements of atoms or groups in space. the phenomenon is called Stereoisomerism (Greek, Stereos = occupying space). The stereoisomers have the same structural formula but differ in the spatial arrangement of atoms or groups in the molecule. In other words, stereoisomerism is exhibited by such compounds which have identical molecular structure but different configurations.
Stereoisomerism is of three types:
a) Geometrical or cis-trans isomerism.
b) Optical Isomerism.
c) Conformational isomerism.
i) Structural Isomerism ii) Stereoisomerism
i) Structural isomerism : When the isomerism is simply due to difference in the arrangement of atoms within the molecule without any reference to space, the phenomenon is termed structural isomerism. In other words, while they have same molecular formulas they possess different structural formula. This type of isomerism which arises from difference in the structure of molecules, includes:
a) Chain or Nuclear Isomerism
b) Positional Isomerism
c) Metamerism
d) Functional Isomerism
e) Ring Chain Isomerism
f) Tautomerism
ii) Stereoisomerism : When isomerism is caused by the different arrangements of atoms or groups in space. the phenomenon is called Stereoisomerism (Greek, Stereos = occupying space). The stereoisomers have the same structural formula but differ in the spatial arrangement of atoms or groups in the molecule. In other words, stereoisomerism is exhibited by such compounds which have identical molecular structure but different configurations.
Stereoisomerism is of three types:
a) Geometrical or cis-trans isomerism.
b) Optical Isomerism.
c) Conformational isomerism.