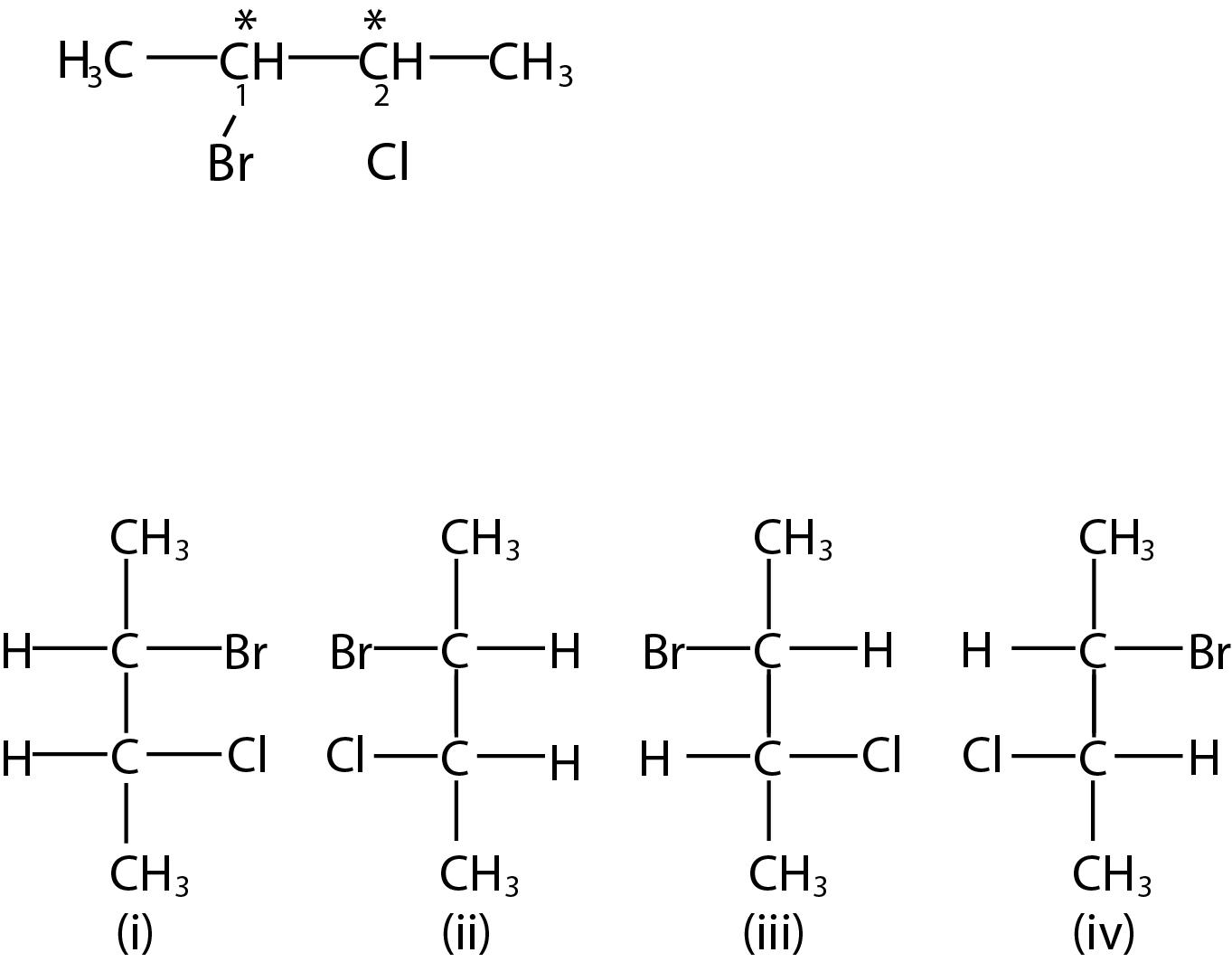
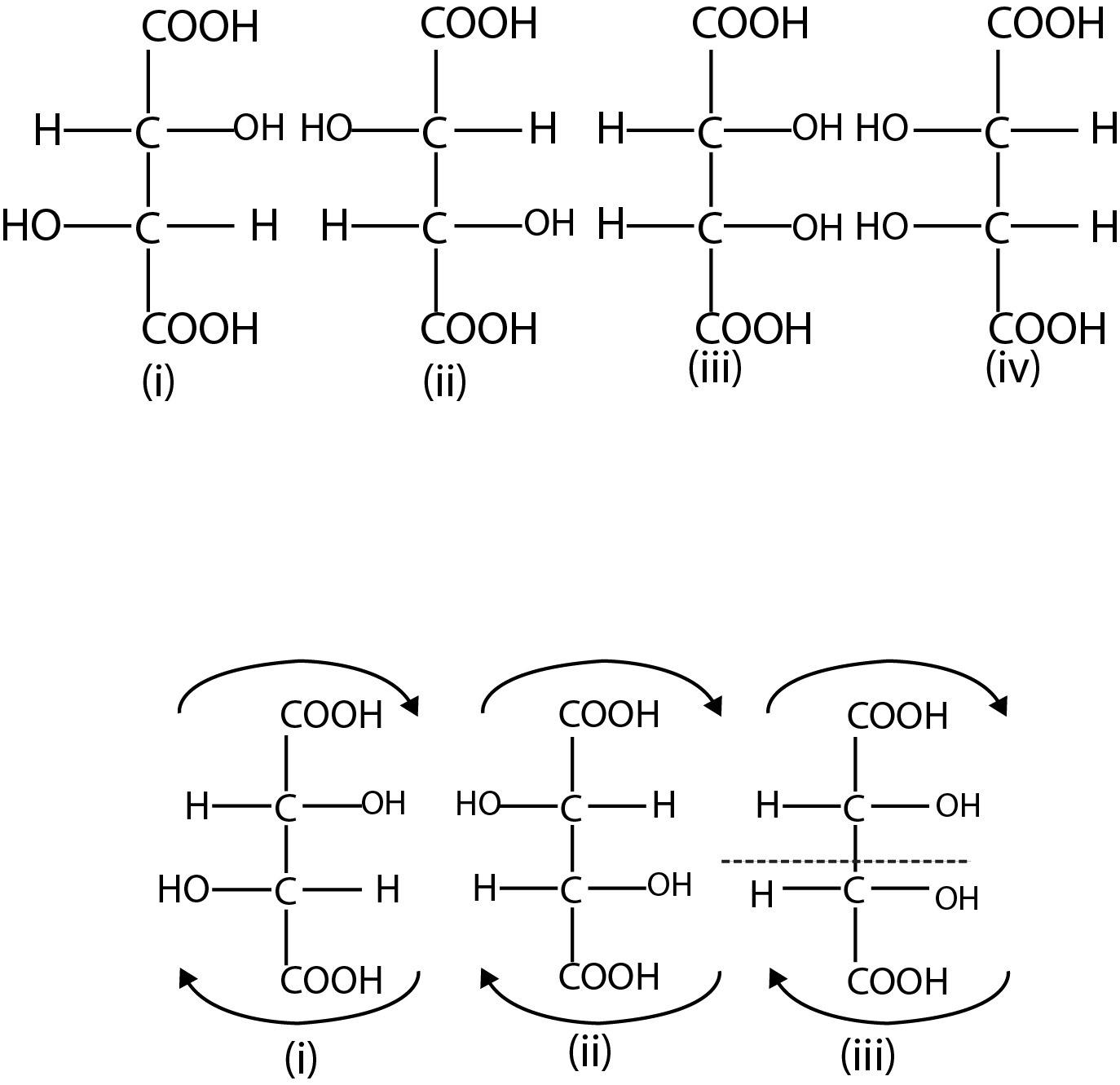
Absolute and Relative Configuration :
While discussing optical isomerism, we must distinguish between relative and absolute configuration (arrangement of atoms or groups) about the asymmetric carbon atom. let us consider a pair of enantiomers, say (+)' and(- )' lactic acid. We know that they differ from one another in the direction in which they rotate the plane of polarised light. In other words, we know their relative configuration in the sense that one is of opposite configuration to the other. But we have no knowledge of the absolute configuration of the either isomer. That is, we cannot tell as to which of the two possible configuration corresponds to {+) - acid and which to the (- ) - acid.
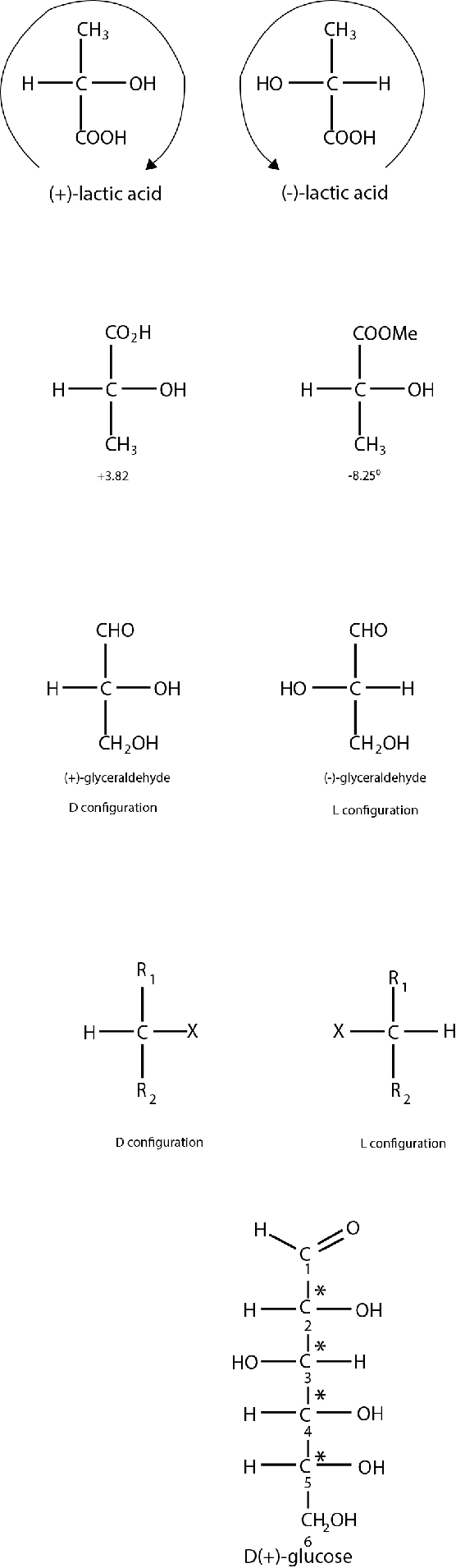
D and L system : The sign of rotation of plane-polarized light by an enantiomer cannot be easily related to either its absolute or relative configuration. Compounds with similar configuration at the asymmetric carbon atom may have opposite sign of rotations and compounds with different configuration may have same sign of rotation. Thus d -lactic acid with a specific rotation + `3.82^(o)` gives I -methyl lactate with a specific rotation `-8.25^(o)`, although the configuration (or arrangement) about the asymmetric carbon atom remains the same during the change. Obviously there appears to be no relation between configuration and sign of rotation. Thus D/L system has been used to specify the configuration at the asymmetric carbon atom. In this system, the configuration of an enantiomer is related to a standard, glyceraldehyde. The two forms of glyceraldehyde were arbitrarily assigned the absolute configurations as shown below.
If the configuration at the asymmetric carbon atom of a compound can be related to `D` (+)-glyceraldehyde, it belongs to `D`- series; and if it can be related to `L`(- )-glyceraldehyde, the compound belongs to `L`-series. Thus many of the naturally occurring a -amino acids have been correlated with glyceraldehyde by chemical transformations. For example, natural alanine {2-aminopropanoic acid) has been elated to `L`(+)-lactic acid which is related to `L`(-)-glyceraldehyde. Alanine, therefore, belongs to the `L`-series In general, the absolute configuration of a substituent (`X`) at the asymmetric centre is specified by writing the projection formula with the carbon chain vertical and the lowest number carbon at the top. The `D` configuration is then the one that has the substituent 'X' on the bond extending to the 'right' of the asymmetric carbon, whereas the `L` configuration has the substituent 'X' on the 'left'. Thus,When there are several asymmetric carbon atoms in a molecule, the configuration at one centre is usually related directly or indirectly to glyceraldehyde, and the configurations at the natural (+)-glucose there are four asymmetric centres (marked by asterisk). By convention for sugars, the
configuration of the highest numbered asymmetric carbon is referred to glyceraldehyde to determine the overall configuration of the molecule. For glucose, this atom is `C- 5` and, therefore, `OH` on it is to the right. Hence the naturally occurring glucose belongs to the `D`-series and is named as `D`- glucose.
R and S System :This is a newer and more systematic method of specifying absolute configuration to optically active compounds. Since it has been proposed by `R,S` Cahn, `C,K` Ingold and `V, P` relog, this system is also known as Cahn-lngold-Prelog system. This system of designating configuration has been used increasingly since the early 1960's and may eventually replace the `DL`-system. Cahn-1 ngold-Prelog system is based on the actual threedimensional or tetrahedral structure of the compound. In order to specify configuration about an asymmetric carbon `C` abde , the groups `a, b, d` and `e` are first assigned an order of priority determined by the 'sequence rules'. These rules will be given later. For the present, let us assign priorities `1, 2, 3, 4` to the groups a, b, d, e. Thus the order of priorities may be stated as
`a > b > d > e`
`(I) (2) (3) (4)`
Now the tetrahedral model of the molecule is viewed from the direction opposite to the group `'e'` of lowest priority `(4)`. The 'conversion rule' says that :
i) If the eye while moving from `a b d` travels in a clockwise or right-hand direction, the configuration is designated `R` (Latin, Rectus = right).
D and L system : The sign of rotation of plane-polarized light by an enantiomer cannot be easily related to either its absolute or relative configuration. Compounds with similar configuration at the asymmetric carbon atom may have opposite sign of rotations and compounds with different configuration may have same sign of rotation. Thus d -lactic acid with a specific rotation + `3.82^(o)` gives I -methyl lactate with a specific rotation `-8.25^(o)`, although the configuration (or arrangement) about the asymmetric carbon atom remains the same during the change. Obviously there appears to be no relation between configuration and sign of rotation. Thus D/L system has been used to specify the configuration at the asymmetric carbon atom. In this system, the configuration of an enantiomer is related to a standard, glyceraldehyde. The two forms of glyceraldehyde were arbitrarily assigned the absolute configurations as shown below.
If the configuration at the asymmetric carbon atom of a compound can be related to `D` (+)-glyceraldehyde, it belongs to `D`- series; and if it can be related to `L`(- )-glyceraldehyde, the compound belongs to `L`-series. Thus many of the naturally occurring a -amino acids have been correlated with glyceraldehyde by chemical transformations. For example, natural alanine {2-aminopropanoic acid) has been elated to `L`(+)-lactic acid which is related to `L`(-)-glyceraldehyde. Alanine, therefore, belongs to the `L`-series In general, the absolute configuration of a substituent (`X`) at the asymmetric centre is specified by writing the projection formula with the carbon chain vertical and the lowest number carbon at the top. The `D` configuration is then the one that has the substituent 'X' on the bond extending to the 'right' of the asymmetric carbon, whereas the `L` configuration has the substituent 'X' on the 'left'. Thus,When there are several asymmetric carbon atoms in a molecule, the configuration at one centre is usually related directly or indirectly to glyceraldehyde, and the configurations at the natural (+)-glucose there are four asymmetric centres (marked by asterisk). By convention for sugars, the
configuration of the highest numbered asymmetric carbon is referred to glyceraldehyde to determine the overall configuration of the molecule. For glucose, this atom is `C- 5` and, therefore, `OH` on it is to the right. Hence the naturally occurring glucose belongs to the `D`-series and is named as `D`- glucose.
R and S System :This is a newer and more systematic method of specifying absolute configuration to optically active compounds. Since it has been proposed by `R,S` Cahn, `C,K` Ingold and `V, P` relog, this system is also known as Cahn-lngold-Prelog system. This system of designating configuration has been used increasingly since the early 1960's and may eventually replace the `DL`-system. Cahn-1 ngold-Prelog system is based on the actual threedimensional or tetrahedral structure of the compound. In order to specify configuration about an asymmetric carbon `C` abde , the groups `a, b, d` and `e` are first assigned an order of priority determined by the 'sequence rules'. These rules will be given later. For the present, let us assign priorities `1, 2, 3, 4` to the groups a, b, d, e. Thus the order of priorities may be stated as
`a > b > d > e`
`(I) (2) (3) (4)`
Now the tetrahedral model of the molecule is viewed from the direction opposite to the group `'e'` of lowest priority `(4)`. The 'conversion rule' says that :
i) If the eye while moving from `a b d` travels in a clockwise or right-hand direction, the configuration is designated `R` (Latin, Rectus = right).