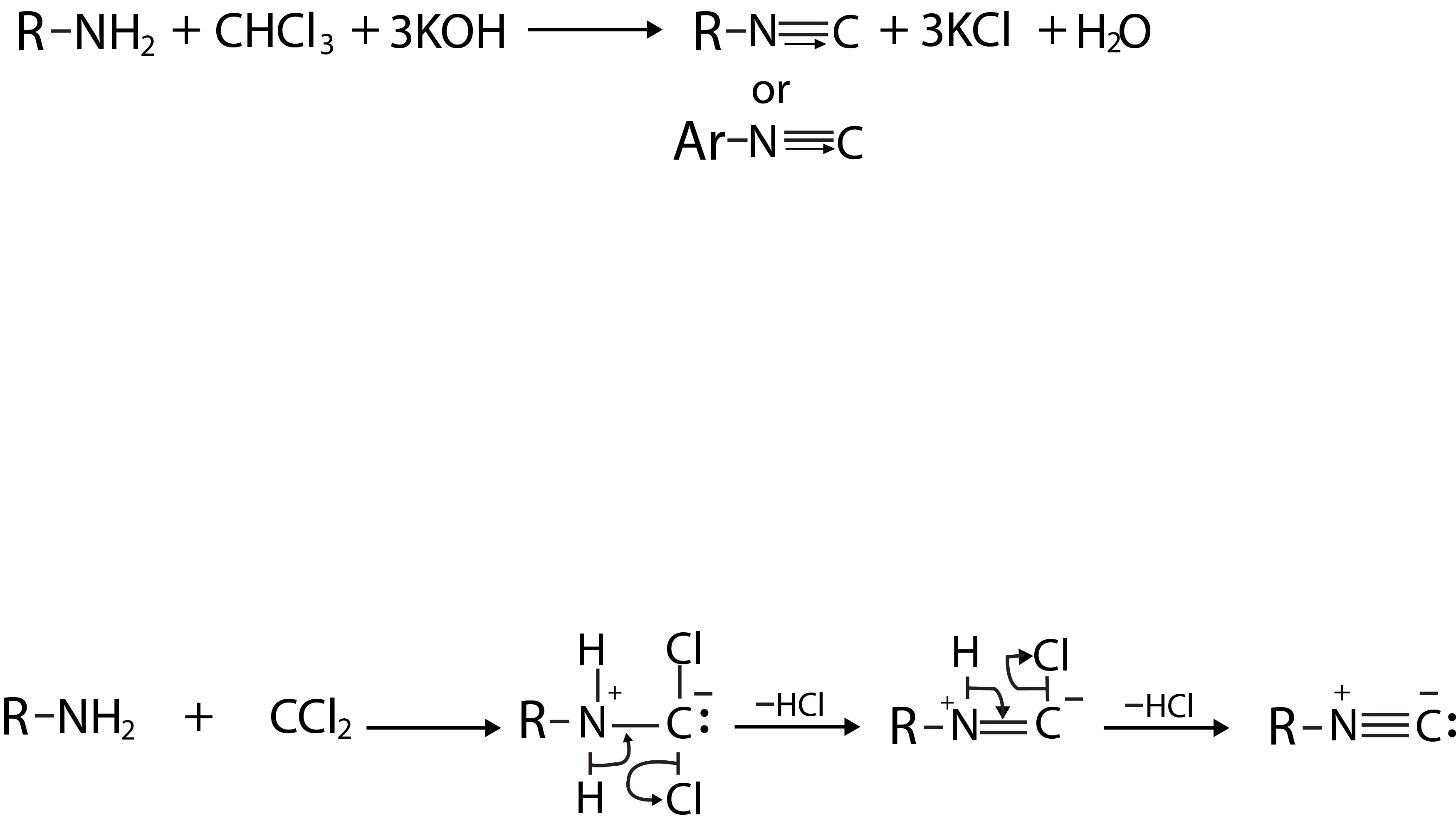
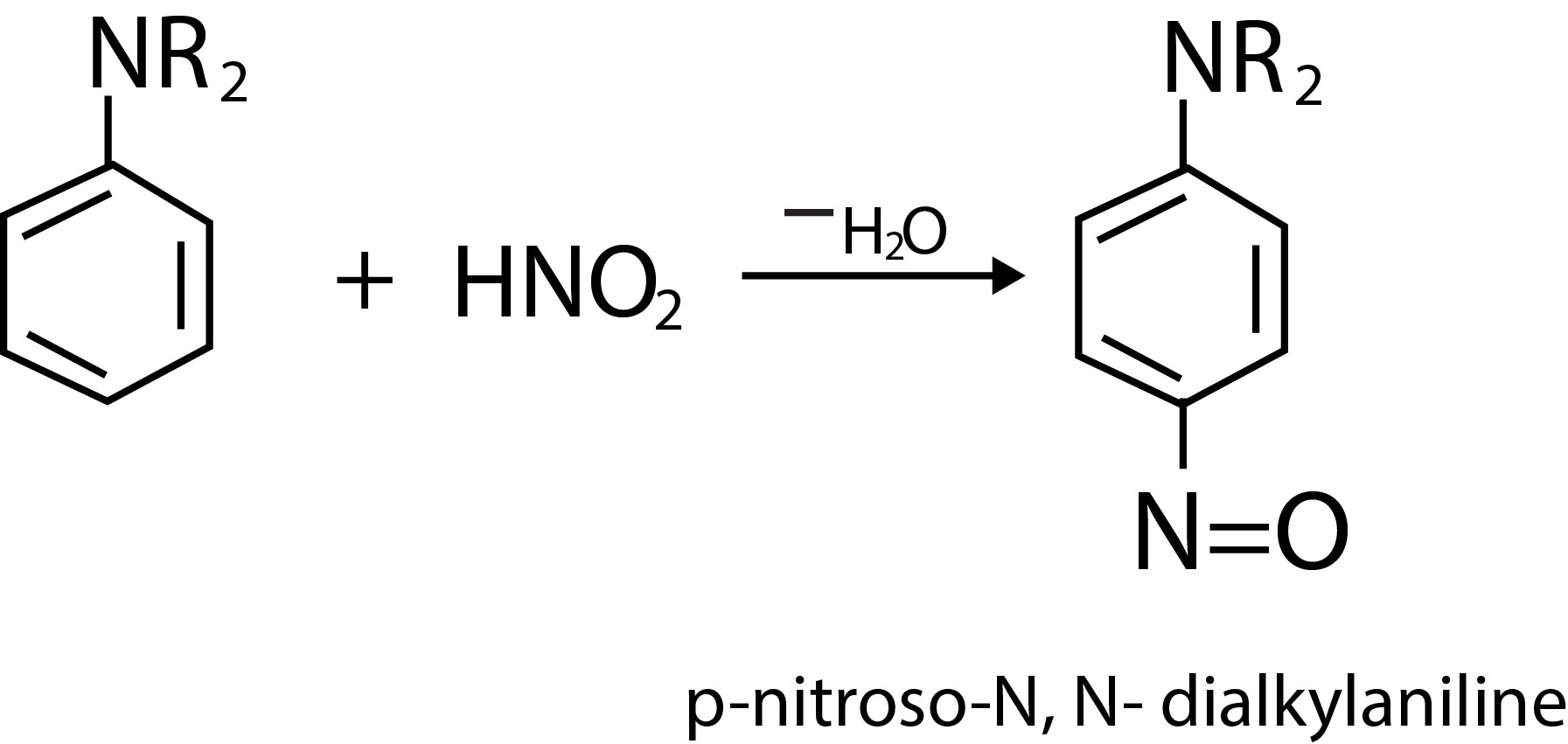
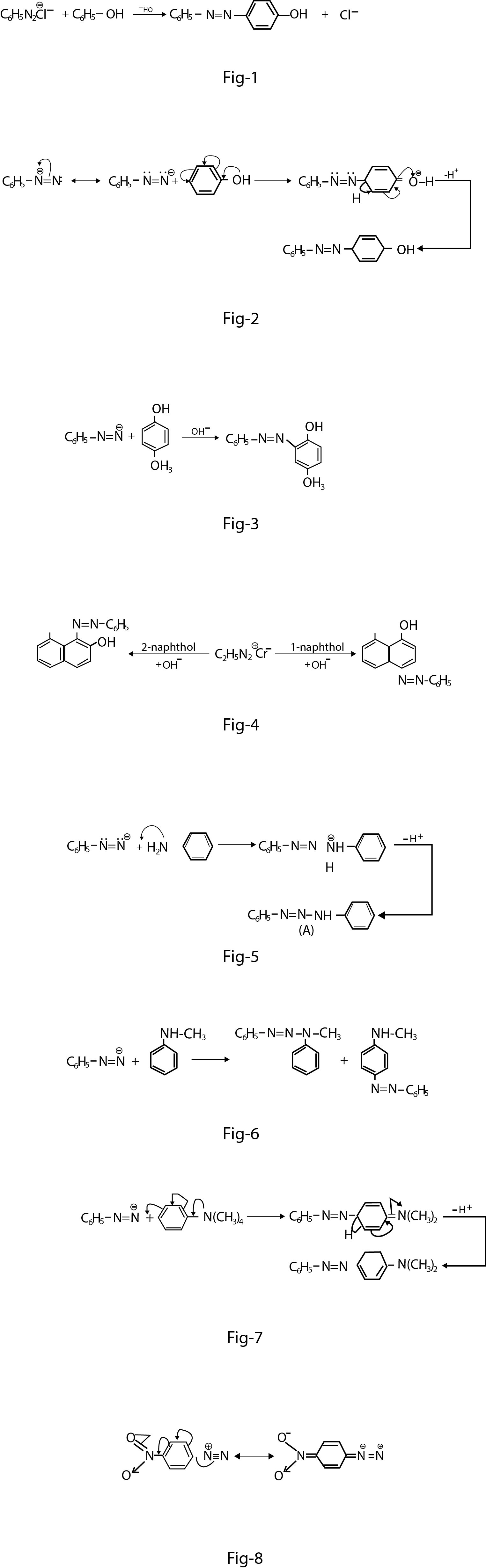
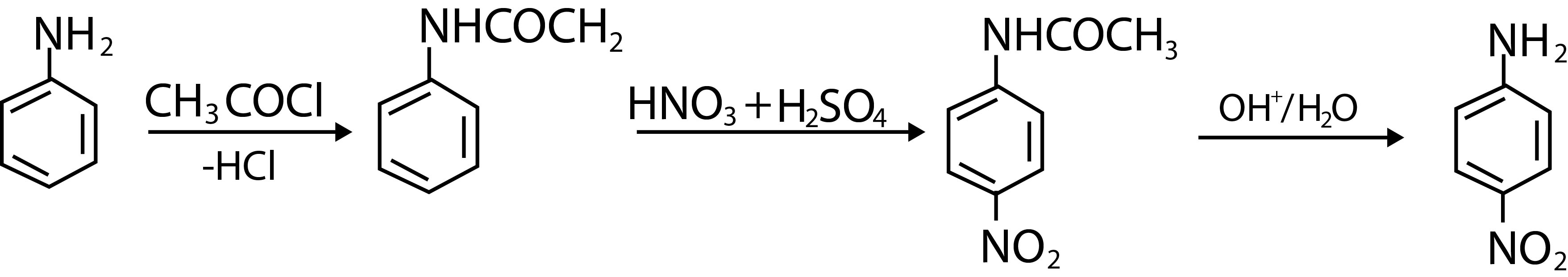
Diazonium salts readily undergo coupling reactions with phenols, naphthols and aromatic amines to form highly coloured azo compounds. For example, benzene diazonium chloride couples with phenol in weakly alkaline solution to form `p`-hydroxyazobenzene(fig.1).
The rate of reaction increases as the `pH` change from `5` to `8` Under mildly alkaline conditions. phenol behaves as phenoxide ion, which is much more activating than phenol itself(fig.2).
Coupling with benzene substrates occurs preferentially in the para position to the hydroxyl group. But if this position is blocked, then the coupling occurs at the ortho position. For example, `p`- cresol gives `o`- azo compound(fig.3).
`1`- and `2`- naphthols in alkaline solution couple with diazonium salt in the `4`- and `1`- position respectively(fig. 4).
Aromatic amines are in general somewhat less readily attacked than phenols and coupling is often carried out in slightly acid solution. Under these conditions not only the concentration of `C_6H_5N_2^(+)` is high but also the amine `ArNH_2` is not significantly converted into the unreactive protonated cation, `Aroverset(+)NH_3`.
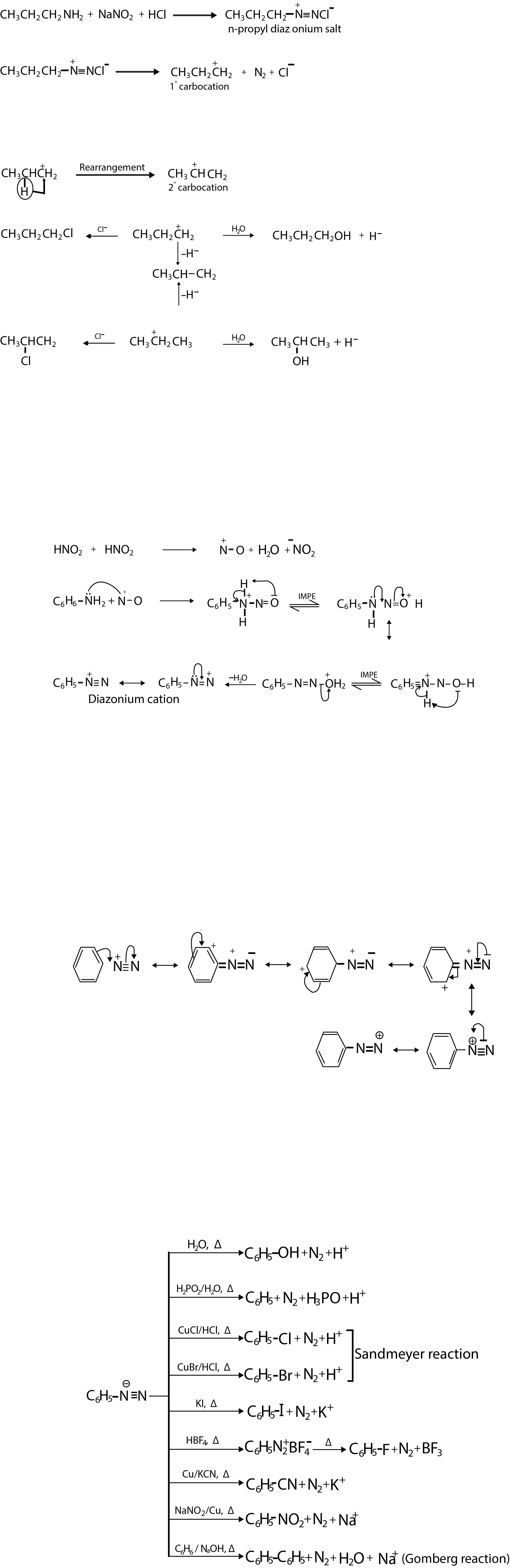
The initial diazotisation of aromatic primary amines is carried out in strongly acidic media to ensure that as yet unreacted amine is converted to the cation and so prevented from coupling with diazonium salt as it is formed.
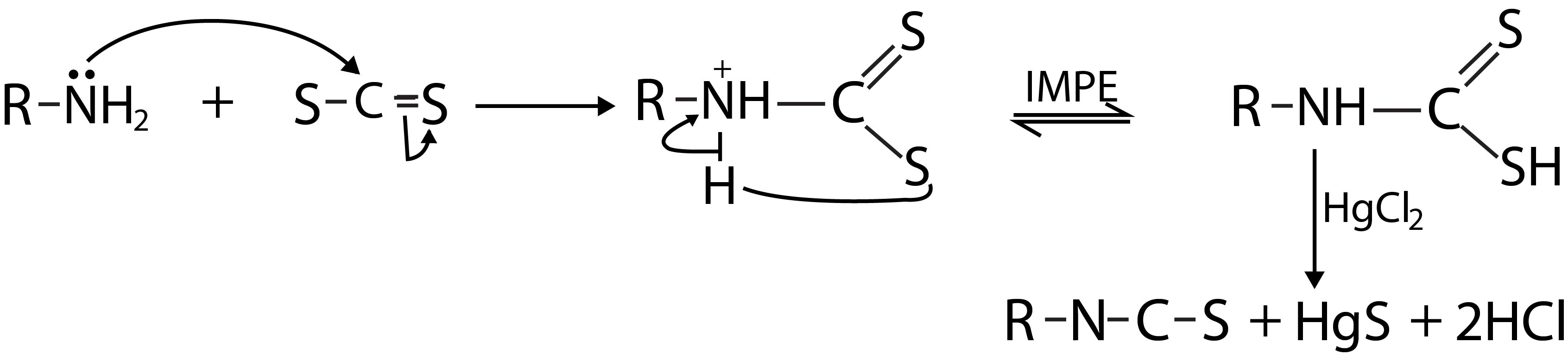
With aromatic amines, there is the possibility of attack on either nitrogen or carbon. In the case of primary amines, the attack of diazonium ion mainly takes place at the nitrogen forming diazo amino compound (`A`)(fig. 5).
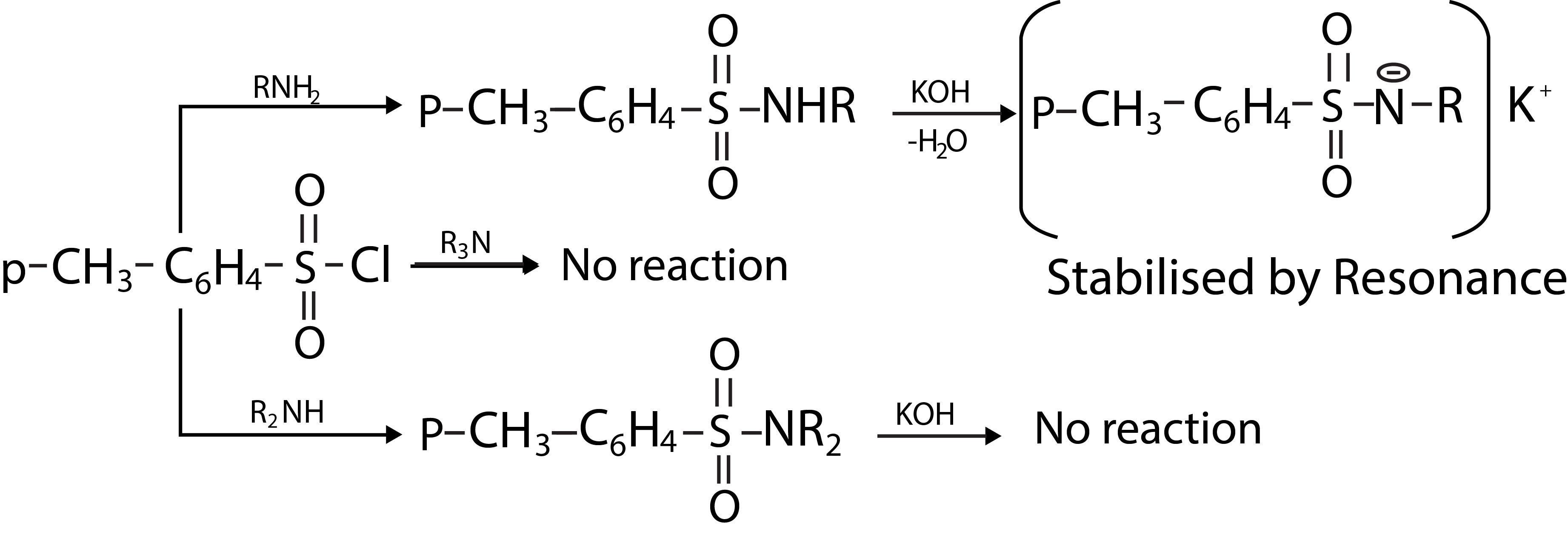
With secondary amines (e.g., `N`- alkyl anilines), two products are formed; one due to `N - N` coupling and the other due to `N - C` coupling. For example, see fig-6.
Tertiary amines (e.g. `N, N`- dialkylaniline) show only `N - C` coupling. Diazonium cation, `C_6H_5N_2^+` is a relatively weak electrophile and reacts with only highly reactive aromatic compounds such as phenols, aniline and substituted anilines(fig.7).
It does not react with less reactive compound `C_5H_5 - OCH_3` (anisole), mesitylene etc. Its reactivity can be increased by introducing strongly electron withdrawing groups such as `NO_2` at the ortho or para position. This will enhance positive charge at the diazo group making it a better electrophiIe(fig.8).
Thus, the `2,4`- dinitrophenyl diazonium cation will react with `C_6H_5 - OCH_3` and `2,4,6`- trinitrophenyl diazonium cation will even react with the hydrocarbon `1 ,3,5`- trimethyl benzene (mesitylene).
Diazonium salts readily undergo coupling reactions with phenols, naphthols and aromatic amines to form highly coloured azo compounds. For example, benzene diazonium chloride couples with phenol in weakly alkaline solution to form `p`-hydroxyazobenzene(fig.1).
The rate of reaction increases as the `pH` change from `5` to `8` Under mildly alkaline conditions. phenol behaves as phenoxide ion, which is much more activating than phenol itself(fig.2).
Coupling with benzene substrates occurs preferentially in the para position to the hydroxyl group. But if this position is blocked, then the coupling occurs at the ortho position. For example, `p`- cresol gives `o`- azo compound(fig.3).
`1`- and `2`- naphthols in alkaline solution couple with diazonium salt in the `4`- and `1`- position respectively(fig. 4).
Aromatic amines are in general somewhat less readily attacked than phenols and coupling is often carried out in slightly acid solution. Under these conditions not only the concentration of `C_6H_5N_2^(+)` is high but also the amine `ArNH_2` is not significantly converted into the unreactive protonated cation, `Aroverset(+)NH_3`.
The initial diazotisation of aromatic primary amines is carried out in strongly acidic media to ensure that as yet unreacted amine is converted to the cation and so prevented from coupling with diazonium salt as it is formed.
With aromatic amines, there is the possibility of attack on either nitrogen or carbon. In the case of primary amines, the attack of diazonium ion mainly takes place at the nitrogen forming diazo amino compound (`A`)(fig. 5).
With secondary amines (e.g., `N`- alkyl anilines), two products are formed; one due to `N - N` coupling and the other due to `N - C` coupling. For example, see fig-6.
Tertiary amines (e.g. `N, N`- dialkylaniline) show only `N - C` coupling. Diazonium cation, `C_6H_5N_2^+` is a relatively weak electrophile and reacts with only highly reactive aromatic compounds such as phenols, aniline and substituted anilines(fig.7).
It does not react with less reactive compound `C_5H_5 - OCH_3` (anisole), mesitylene etc. Its reactivity can be increased by introducing strongly electron withdrawing groups such as `NO_2` at the ortho or para position. This will enhance positive charge at the diazo group making it a better electrophiIe(fig.8).
Thus, the `2,4`- dinitrophenyl diazonium cation will react with `C_6H_5 - OCH_3` and `2,4,6`- trinitrophenyl diazonium cation will even react with the hydrocarbon `1 ,3,5`- trimethyl benzene (mesitylene).