-It is delocalisation of sigma electron.
-Also known as sigma-pi-conjugation or no bond resonance.
`text(Occurrence)` :
Alkene, alkynes
Free radicals (saturated type) carbonium ions (saturated type)
`text(Condition :)`
Presence of a `-H` with respect to double bond, triple bond carbon containing positive charge (in carbonium ion) or unpaired electron (in free radicals)
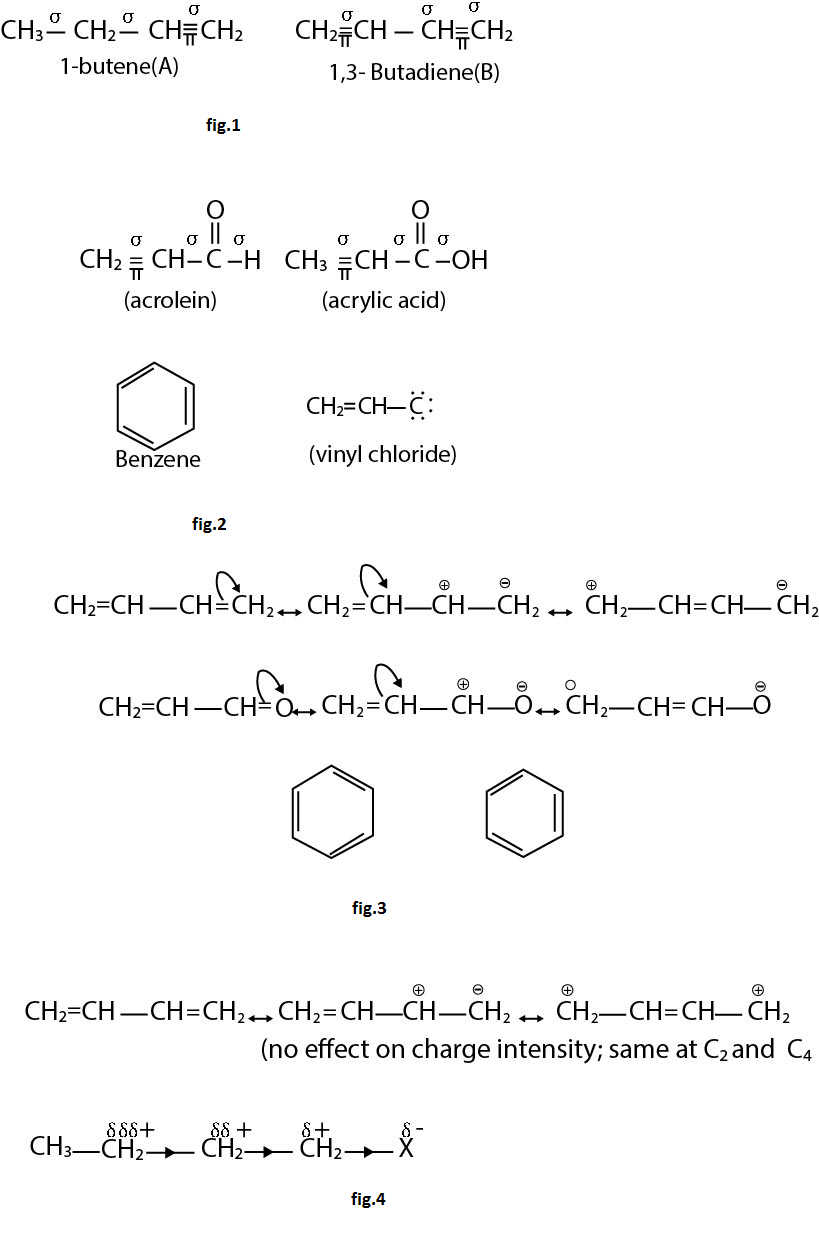
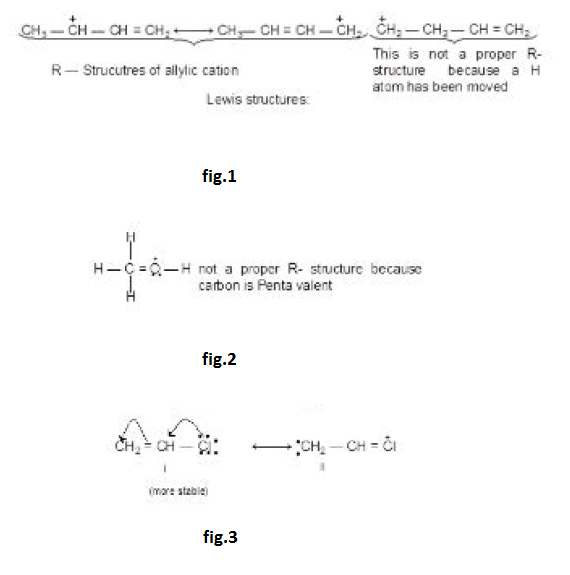
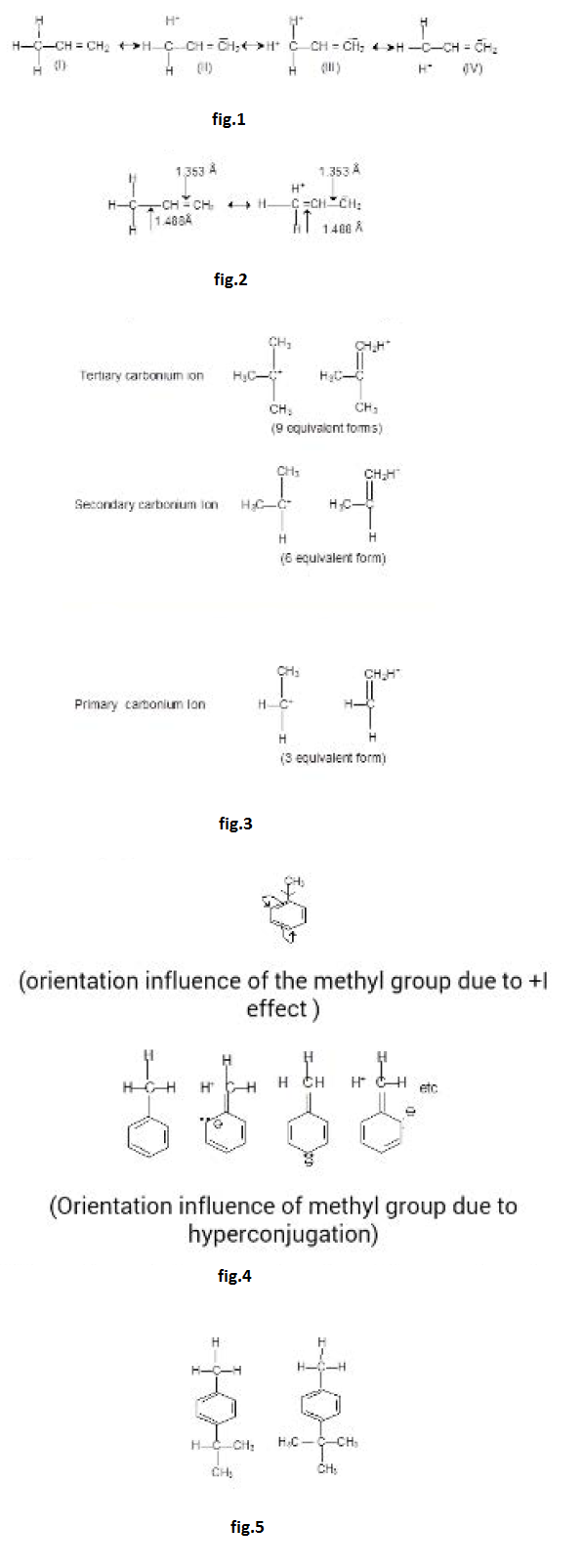
`text(Example :)` See fig.1.
`text(Note :)` Number of hyperconjugative structures = number of `alpha`-Hydrogen. Hence, in above examples structures i, ii, iii, iv are hyperconjugate structures (H-structures).
Hyperconjugation is a permanent effect.
`text(Effects of hyperconjugation :)`
i) `text(Bond Length :)`
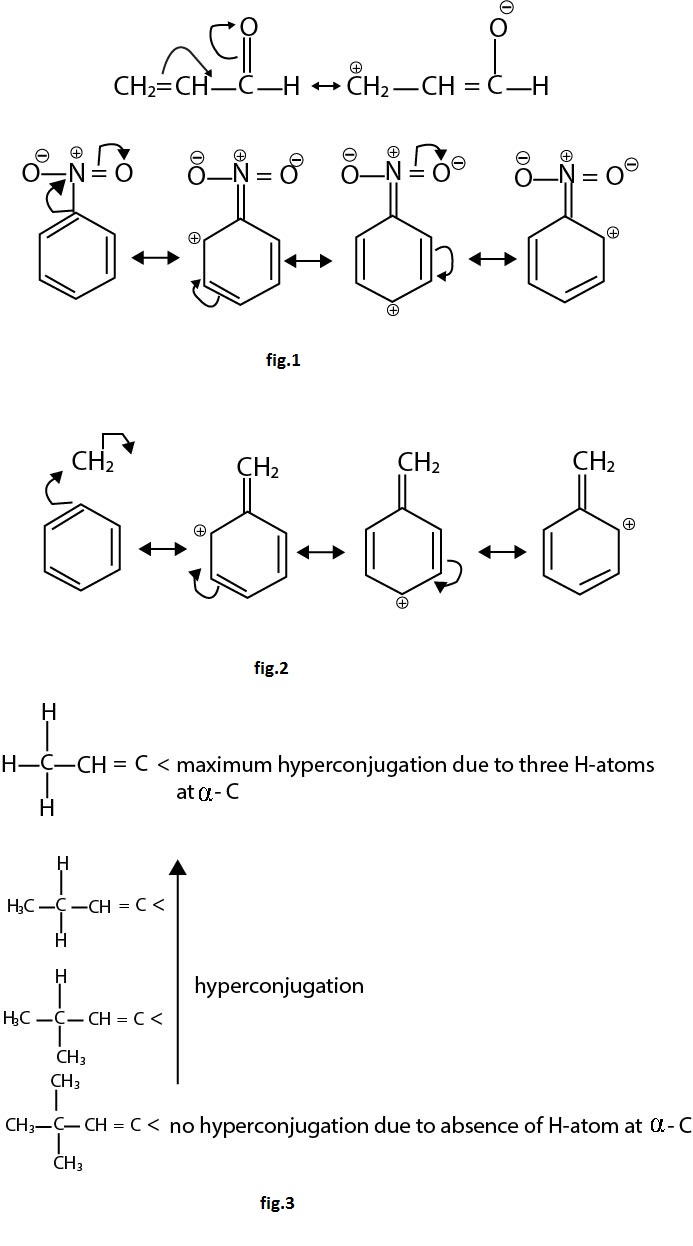
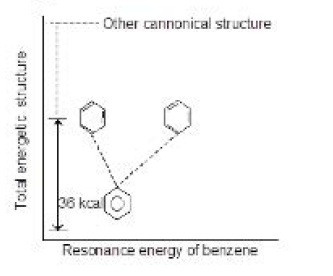
Like resonance, hyperconjugation also affects bond lengths because during the process the single bond in compound acquires some double bond character and vice-versa. Eg. `C-C` bond length in propene is `1.488 A^o` as compared to `1.334 A^o` in ethylene. See fig.2.
ii) `text(Dipole moment :)`
Since hyperconjugation causes these development of charges, it also affects the dipole moment of the molecule..
iii) `text(Stability of carbonium Ions :)`
The order of stability of carbonium ions is as follows :
Tertiary > Secondary > Primary.
Above order of stability can be explained by hyperconjugation. In general the number of hydrogen atoms attached to a carbon atoms, the more hyperconjugtaion forms can be written and thus greater will be the stability of carbonium ions. See fig.3.
iv) `text(Stability of Free radicals :)`
Stability of Free radicals can also be explained as that of carbonium ion.
`(CH_2)overset(.)(C) > (CH_2)_2overset(.)CH > CH_2overset(.)CH_2 > overset(.)CH_2`
v) `text(Orientation influence of methyl group :)`
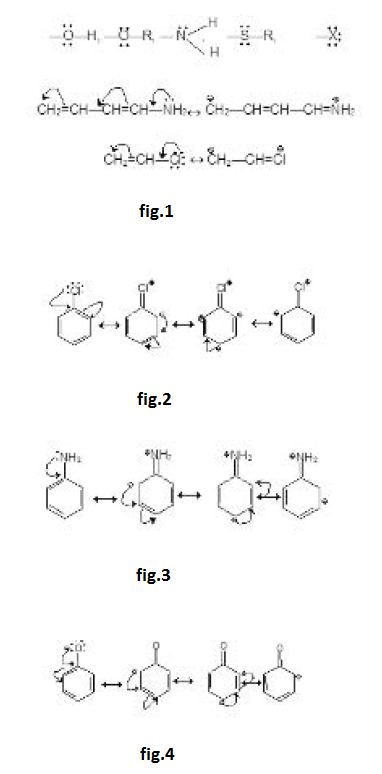
The o, p-directing influence of the methyl group in methyl benzenes is attributed partly to inductive and party of hyperconjugation effect. See fig.4.
The role of hyperconjugation in o,p-directing influence of methyl group is evidenced by the part that nitration of p-iso propyl toluene and p-tert-butyl toluene form the product in which `-NO_2` group is introduced in the ortho position with respect to methyl group and not to isopropyl or t-butyl group although the latter groups are more electron donating than Methyl groups. See fig.5.
i.e., The substitution takes place contrary to inductive effect. Actually this constitutes an example where hyperconjugation overpowers inductive effect.
-It is delocalisation of sigma electron.
-Also known as sigma-pi-conjugation or no bond resonance.
`text(Occurrence)` :
Alkene, alkynes
Free radicals (saturated type) carbonium ions (saturated type)
`text(Condition :)`
Presence of a `-H` with respect to double bond, triple bond carbon containing positive charge (in carbonium ion) or unpaired electron (in free radicals)
`text(Example :)` See fig.1.
`text(Note :)` Number of hyperconjugative structures = number of `alpha`-Hydrogen. Hence, in above examples structures i, ii, iii, iv are hyperconjugate structures (H-structures).
Hyperconjugation is a permanent effect.
`text(Effects of hyperconjugation :)`
i) `text(Bond Length :)`
Like resonance, hyperconjugation also affects bond lengths because during the process the single bond in compound acquires some double bond character and vice-versa. Eg. `C-C` bond length in propene is `1.488 A^o` as compared to `1.334 A^o` in ethylene. See fig.2.
ii) `text(Dipole moment :)`
Since hyperconjugation causes these development of charges, it also affects the dipole moment of the molecule..
iii) `text(Stability of carbonium Ions :)`
The order of stability of carbonium ions is as follows :
Tertiary > Secondary > Primary.
Above order of stability can be explained by hyperconjugation. In general the number of hydrogen atoms attached to a carbon atoms, the more hyperconjugtaion forms can be written and thus greater will be the stability of carbonium ions. See fig.3.
iv) `text(Stability of Free radicals :)`
Stability of Free radicals can also be explained as that of carbonium ion.
`(CH_2)overset(.)(C) > (CH_2)_2overset(.)CH > CH_2overset(.)CH_2 > overset(.)CH_2`
v) `text(Orientation influence of methyl group :)`
The o, p-directing influence of the methyl group in methyl benzenes is attributed partly to inductive and party of hyperconjugation effect. See fig.4.
The role of hyperconjugation in o,p-directing influence of methyl group is evidenced by the part that nitration of p-iso propyl toluene and p-tert-butyl toluene form the product in which `-NO_2` group is introduced in the ortho position with respect to methyl group and not to isopropyl or t-butyl group although the latter groups are more electron donating than Methyl groups. See fig.5.
i.e., The substitution takes place contrary to inductive effect. Actually this constitutes an example where hyperconjugation overpowers inductive effect.