Temperature :
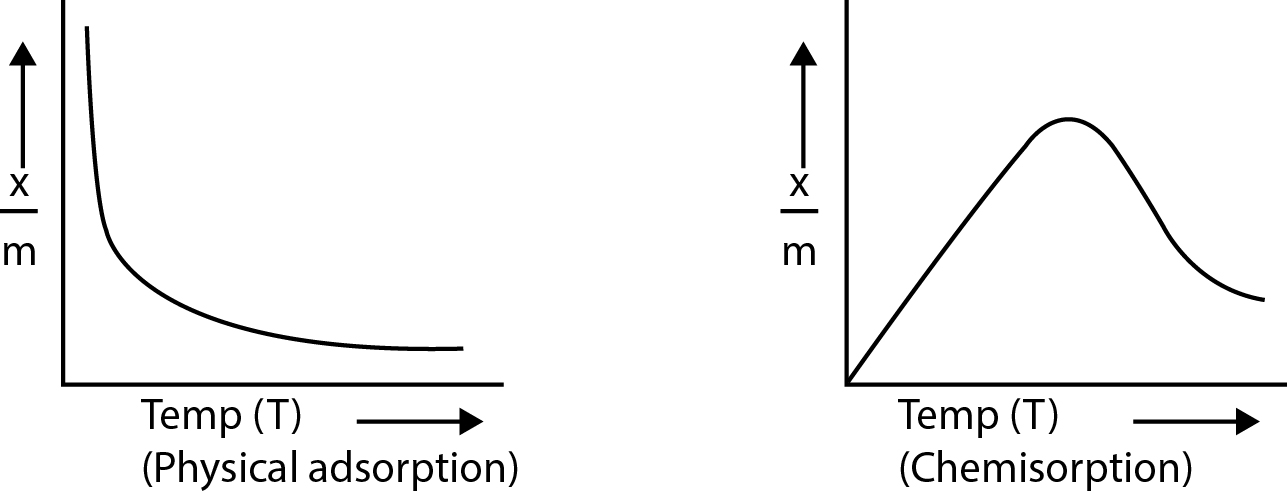
As adsorption is accompanied by release of heat energy, so in accordance with Le . Chatelier's principle, the increase of temperature should decrease the extent of adsorption. This has indeed been found to be so. A plot of `x //m` vs temperature at constant pressure is called `text(adsorption isobar)` . In the case of physical adsorption `x //m` decreases with increase of temperature whereas in the case of
chemisorption, `x //m` initially increases with temperature and then decreases as shown below. The initial increase is due to the fact that
chemisorption requires activation energy.
chemisorption, `x //m` initially increases with temperature and then decreases as shown below. The initial increase is due to the fact that
chemisorption requires activation energy.