Electrophoresis or Cataphoresis :
The particles of the colloidal solution carry same type of charge, either positive or negative. The dispersion medium carries an equal and opposite charge. The colloidal solutions as a whole are electrically neutral. The origin of electrical charge on colloidal particles could be due to frictional electrification, electron captures or preferential adsorption of ions from solutions depending on the method used for the preparation of colloidal solutions. Due to similar nature of the charge carried by the particles, they repel each other and do not combine to form aggregates. This makes a colloidal solution stable and the colloidal particles do not settle down. Some of the common positively and negatively charged colloids are given below:
`text(Positively charged)`
`Fe(OH)_3` sol, `Cr(OH)_3` sol, `Al(OH)_3` sol, `Ca(OH)_2`, `TiO_2`, dyes like methylene blue and haemoglobin.
`text(Negatively charged)`
`As_2S_3` sol, `Sb_2S_3` sol, `CdS` sol, `Au` sol, `Cu` sol, `Ag` sol and acid dyes like congo red.
The existence of charge is shown by passing electric current through two electrodes when all the colloidal particles move towards the same electrode either cathode or anode. Positively charged colloidal particles move towards negatively charged cathode whereas negatively charged colloidal particles move towards positively charged anode. The movement of colloidal particles under the influence of electric field is called electrophoresis or cataphoresis.
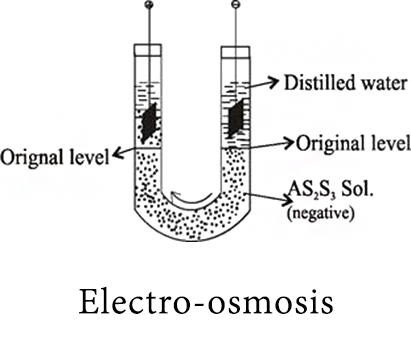
`text(Electro-osmosis)` : If the dispersed phase is prevented from moving, the application of an EMF will, however result in movement of the dispersion medium. This is the basis of the phenomenon of electro-osmosis i.e., the passage of dispersion medium of a colloid through a porous diaphragm under the influence of an applied electric field. This can be demonstrated in the following way. In the figure given, the plug of clay provides stationary negatively charged colloidal particles. Water, in contact with clay, must therefore carry a positive charge. Under the influence of an electric field, water will be found to move towards the cathode.
`text(Positively charged)`
`Fe(OH)_3` sol, `Cr(OH)_3` sol, `Al(OH)_3` sol, `Ca(OH)_2`, `TiO_2`, dyes like methylene blue and haemoglobin.
`text(Negatively charged)`
`As_2S_3` sol, `Sb_2S_3` sol, `CdS` sol, `Au` sol, `Cu` sol, `Ag` sol and acid dyes like congo red.
The existence of charge is shown by passing electric current through two electrodes when all the colloidal particles move towards the same electrode either cathode or anode. Positively charged colloidal particles move towards negatively charged cathode whereas negatively charged colloidal particles move towards positively charged anode. The movement of colloidal particles under the influence of electric field is called electrophoresis or cataphoresis.
`text(Electro-osmosis)` : If the dispersed phase is prevented from moving, the application of an EMF will, however result in movement of the dispersion medium. This is the basis of the phenomenon of electro-osmosis i.e., the passage of dispersion medium of a colloid through a porous diaphragm under the influence of an applied electric field. This can be demonstrated in the following way. In the figure given, the plug of clay provides stationary negatively charged colloidal particles. Water, in contact with clay, must therefore carry a positive charge. Under the influence of an electric field, water will be found to move towards the cathode.