Calculation of Work Done in Various Process :
(i) `text(Isothermal process : In isothermal process, work done can be calculated as)`
`w = - int_(V_1)^(V_2) PdV`
Since `dT =0 => dU =0` for an ideal gas
from 1st law `q = -w`
(a) If process is reversible
`w = -n RT ln (V_2/V_1)`
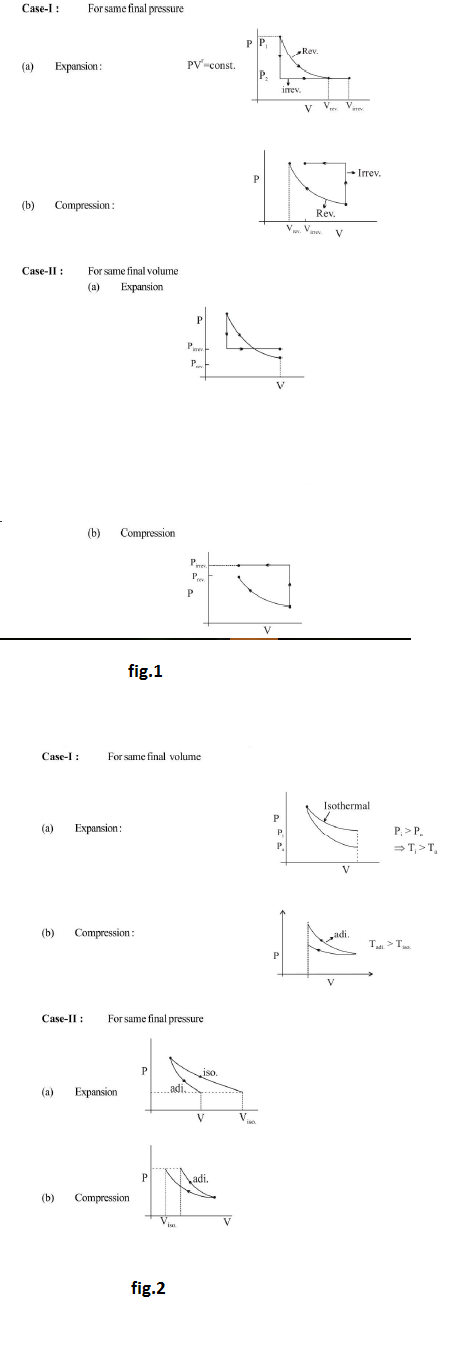
(b) `text(Irreversible isothermal expansion)` : If external pressure over the piston is abruptly changed from the equilibrium value, the mechanical equilibrium of system is disturbed and piston rushes out :
This type of `PV` work is irreversible `PV` work. To calculate irreversible `PV` work, Law of conservation of energy is used. Suppose as a result of difference in pressure a piston moves out and acquire kinetic energy `DeltaKE` and in the process volume increase by `DeltaV` then `W_(irr) = - P_(ext) DeltaV - DeltaKE`
If after sufficient times piston come back to equilibrium state (off course in the process it moves up and down from equilibrium position many times), `DeltaKE = 0` : All the acquired kinetic energy is transferred back to ideal gas. See fig.1.
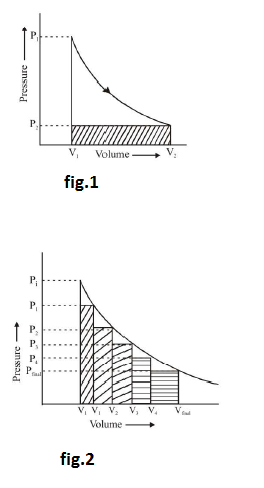
(c) `text[Irreversible isothermal expansion and compression (Many steps)]` : Consider an irreversible expansion of an ideal gas from initial pressure `P_1` to final pressure `P_1` in four steps. The gas is allowed to expand against constant external pressure of `P_1`, `P_2`, `P_3` and `P_4` and finally `P_1`. Hence the system passes on to final state through four equilibrium states. The work done in the process is shown graphically. The area under the isotherm is the magnitude of reversible work. Clearly the magnitude of reversible work of expansion is greater than irreversible work. As the number of intermediate steps in irreversible expansion is increased, the magnitude of work increases, and as number of steps tend to infinity `w_(irr)` tends to `w_(rev)`. The graphical comparison of irreversible and reversible work is shown in fig.2.
(d) `text(Free expansion of ideal gas)` : When ideal gas is allowed to expand against zero external pressure, the process is called free expansion. `w = 0` for free expansion. During the free expansion, the ideal gas do not lose any energy, and hence temperature of ideal gas remains constant. Hence, free expansion of ideal gas is an example of isothermal, adiabatic irreversible process.
However if a real gas is allowed to expand in vacuum, the gas maybe cooled or heated up depending upon temperature of the real gas. The temperature above which a gas gets heated up upon expansion is called inversion temperature.
`text(Important points)` : If the reversible isothermal expansion is reversed by gradually increasing the pressure the system will return to initial state retracing it's path. This means path of reversible process can be exactly reversed if conditions are reversed. Work done by the system during reversible isothermal expansion is maximum possible work obtainable from system under similar condition.
(ii) `text(Isobaric process)` : In isobaric process, pressure remains constant during the process.
`w= int_(V_2)^(V_1) PdV = -P_text(ext.)(V_2 -V_1) = -nRDeltaT` & `DeltaH = q_p`
(iii) `text(lsochoric process)` : In isochoric process, volume remains constant during the process.
Since `dV = 0` `=>` `w=0`
From 1st law `DeltaU = q`
`w = - int_(V_1)^(V_2) PdV`
Since `dT =0 => dU =0` for an ideal gas
from 1st law `q = -w`
(a) If process is reversible
`w = -n RT ln (V_2/V_1)`
(b) `text(Irreversible isothermal expansion)` : If external pressure over the piston is abruptly changed from the equilibrium value, the mechanical equilibrium of system is disturbed and piston rushes out :
This type of `PV` work is irreversible `PV` work. To calculate irreversible `PV` work, Law of conservation of energy is used. Suppose as a result of difference in pressure a piston moves out and acquire kinetic energy `DeltaKE` and in the process volume increase by `DeltaV` then `W_(irr) = - P_(ext) DeltaV - DeltaKE`
If after sufficient times piston come back to equilibrium state (off course in the process it moves up and down from equilibrium position many times), `DeltaKE = 0` : All the acquired kinetic energy is transferred back to ideal gas. See fig.1.
(c) `text[Irreversible isothermal expansion and compression (Many steps)]` : Consider an irreversible expansion of an ideal gas from initial pressure `P_1` to final pressure `P_1` in four steps. The gas is allowed to expand against constant external pressure of `P_1`, `P_2`, `P_3` and `P_4` and finally `P_1`. Hence the system passes on to final state through four equilibrium states. The work done in the process is shown graphically. The area under the isotherm is the magnitude of reversible work. Clearly the magnitude of reversible work of expansion is greater than irreversible work. As the number of intermediate steps in irreversible expansion is increased, the magnitude of work increases, and as number of steps tend to infinity `w_(irr)` tends to `w_(rev)`. The graphical comparison of irreversible and reversible work is shown in fig.2.
(d) `text(Free expansion of ideal gas)` : When ideal gas is allowed to expand against zero external pressure, the process is called free expansion. `w = 0` for free expansion. During the free expansion, the ideal gas do not lose any energy, and hence temperature of ideal gas remains constant. Hence, free expansion of ideal gas is an example of isothermal, adiabatic irreversible process.
However if a real gas is allowed to expand in vacuum, the gas maybe cooled or heated up depending upon temperature of the real gas. The temperature above which a gas gets heated up upon expansion is called inversion temperature.
`text(Important points)` : If the reversible isothermal expansion is reversed by gradually increasing the pressure the system will return to initial state retracing it's path. This means path of reversible process can be exactly reversed if conditions are reversed. Work done by the system during reversible isothermal expansion is maximum possible work obtainable from system under similar condition.
(ii) `text(Isobaric process)` : In isobaric process, pressure remains constant during the process.
`w= int_(V_2)^(V_1) PdV = -P_text(ext.)(V_2 -V_1) = -nRDeltaT` & `DeltaH = q_p`
(iii) `text(lsochoric process)` : In isochoric process, volume remains constant during the process.
Since `dV = 0` `=>` `w=0`
From 1st law `DeltaU = q`