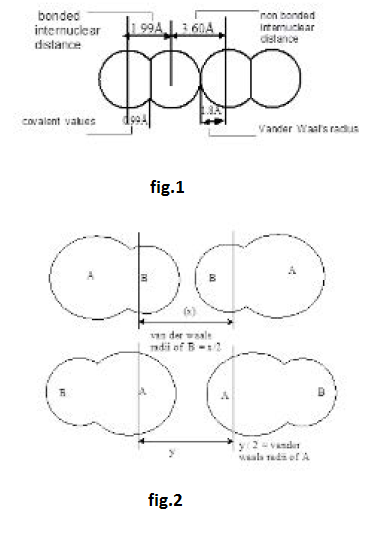
A neutural atom changes to "cation by the Ioss of one or more electrons and to an anion by the gain of one or more electrons. The number of charge on cation and anion is equal to the number of electrons lost or gained respectively. The ionic radii of the ions present in an ionic crystal may be calculated from the inter-nuclear distance between the two ions.
i) `text(Radius of a Cation)` : Radius of a cation is invariably smaller than that of the corresponding neutral atom
`quad quad quad quad Na quad Na^(+)`
Number of `e^(-)= 11 quad 10`
Number of `p = 11 quad 11`
`1 s^2 quad 2s^2 quad 2p^6 quad 3s^1 quad 1 s^2 quad 2s^2 quad 2p^6`
`text(Reasons)` :
a) The effective nuclear charge increases. For example in `Na` atom `11` electrons are attracted by `11` protons and in `Na^(+)`, `10` electrons are attracted by `11` protons. Thus in the formation of cation number of electrons decreases and nuclear charge remains the same.
b) Generally the formation of cation results in the removal of the whole outer shell.
c) Inter electronic repulsion decreases. The inter electronic repulsion in `Na` is among `11e^(-)` and in `Na^(+)` among `10e^(-)`
ii) `text(Radius of an anion)` : Radius of an anion is invariably bigger than that of the corresponding atom.
`quad quad quad quad quad Cl quad Cl^(-)`
Number of `e^(-) = 17 quad 18`
Number of `p = 17 quad 17`
`text(Reasons)` :
a) The effective nuclear charge decrease in the formation of anion. Thus the electrostatic force of attraction between the nucleus and the outer electrons decreases and the size of the anion increases.
b) Inter electronic repulsion increases.
iii) `text(lso-electronic series)` : A series of atoms, ions and molecules in which each species contains same number of electrons but different nuclear charge is called isoelectronic series.
`quad quad quad quad quad quadN^(3-)quad O^(2-) quad F^(-) quad Ne quad Na^(+) quad Mg^(2+)`
Number of `e^(-) quad = 10 quad 10 quad 10 quad 10 quad 10 quad 10`
Number of `p = 7 quad 8 quad 9 quad 10 quad 11 quad 12`
a) Number of electrons is same.
b) Number of protons is increasing
c) So the effective nuclear charge is increasing and atomic size is decreasing. In an iso-electronic series atomic size decreases with the increase of charge.
Some of the examples of iso-electronic series are as under
i) `S^(2-), Cl, K^(+), Ca^(2+), Sc^(3+)`
ii) `SO_2, NO_3^(-), CO_3^(2-)`
iii) `N_2, CO, CN^(-)`
iv) `NH_3, H_3O^(+)`
A neutural atom changes to "cation by the Ioss of one or more electrons and to an anion by the gain of one or more electrons. The number of charge on cation and anion is equal to the number of electrons lost or gained respectively. The ionic radii of the ions present in an ionic crystal may be calculated from the inter-nuclear distance between the two ions.
i) `text(Radius of a Cation)` : Radius of a cation is invariably smaller than that of the corresponding neutral atom
`quad quad quad quad Na quad Na^(+)`
Number of `e^(-)= 11 quad 10`
Number of `p = 11 quad 11`
`1 s^2 quad 2s^2 quad 2p^6 quad 3s^1 quad 1 s^2 quad 2s^2 quad 2p^6`
`text(Reasons)` :
a) The effective nuclear charge increases. For example in `Na` atom `11` electrons are attracted by `11` protons and in `Na^(+)`, `10` electrons are attracted by `11` protons. Thus in the formation of cation number of electrons decreases and nuclear charge remains the same.
b) Generally the formation of cation results in the removal of the whole outer shell.
c) Inter electronic repulsion decreases. The inter electronic repulsion in `Na` is among `11e^(-)` and in `Na^(+)` among `10e^(-)`
ii) `text(Radius of an anion)` : Radius of an anion is invariably bigger than that of the corresponding atom.
`quad quad quad quad quad Cl quad Cl^(-)`
Number of `e^(-) = 17 quad 18`
Number of `p = 17 quad 17`
`text(Reasons)` :
a) The effective nuclear charge decrease in the formation of anion. Thus the electrostatic force of attraction between the nucleus and the outer electrons decreases and the size of the anion increases.
b) Inter electronic repulsion increases.
iii) `text(lso-electronic series)` : A series of atoms, ions and molecules in which each species contains same number of electrons but different nuclear charge is called isoelectronic series.
`quad quad quad quad quad quadN^(3-)quad O^(2-) quad F^(-) quad Ne quad Na^(+) quad Mg^(2+)`
Number of `e^(-) quad = 10 quad 10 quad 10 quad 10 quad 10 quad 10`
Number of `p = 7 quad 8 quad 9 quad 10 quad 11 quad 12`
a) Number of electrons is same.
b) Number of protons is increasing
c) So the effective nuclear charge is increasing and atomic size is decreasing. In an iso-electronic series atomic size decreases with the increase of charge.
Some of the examples of iso-electronic series are as under
i) `S^(2-), Cl, K^(+), Ca^(2+), Sc^(3+)`
ii) `SO_2, NO_3^(-), CO_3^(2-)`
iii) `N_2, CO, CN^(-)`
iv) `NH_3, H_3O^(+)`