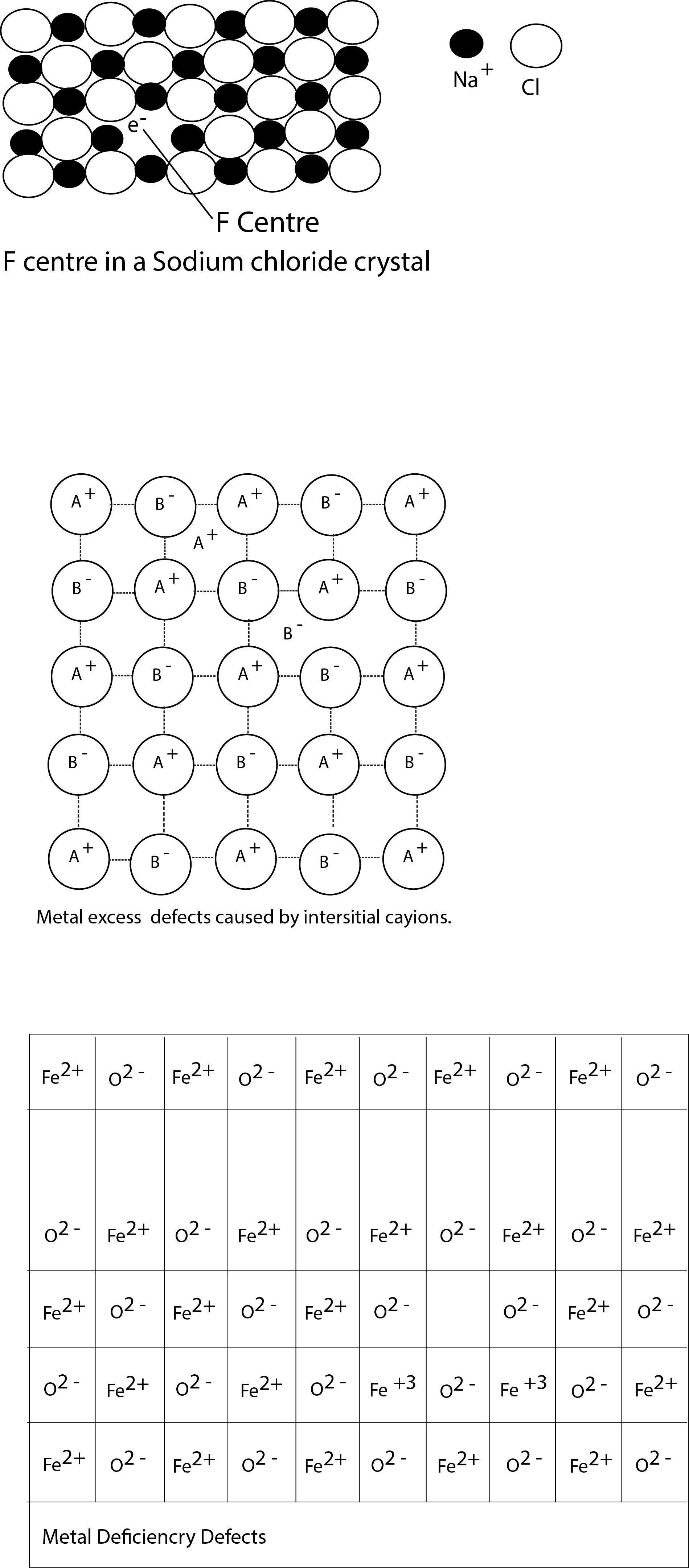
Stoichiometric Defects :
Stoichiometric compounds are those where the numbers of the different types of atoms or ions present are exactly in the ratios indicated by their chemical formulae.They obey the law of constant composition that the same chemical compound always contains the same elements in the same composition by weight. Two types of defects may be observed in stoichiometric compounds, called Schottky and Frenkel defects respectively. At absolute zero, crystals tend to have a perfectly ordered arrangement. As the temperature increases the amount of thermal vibration of ions in their lattice sites increases and if the vibration of a particular ion becomes large enough, it may jump out of its lattice site. The higher the temperature, the greater the chance that lattice sites may be unoccupied. Since the number of defects depends on the temperature, these are sometimes called thermodynamic effect.
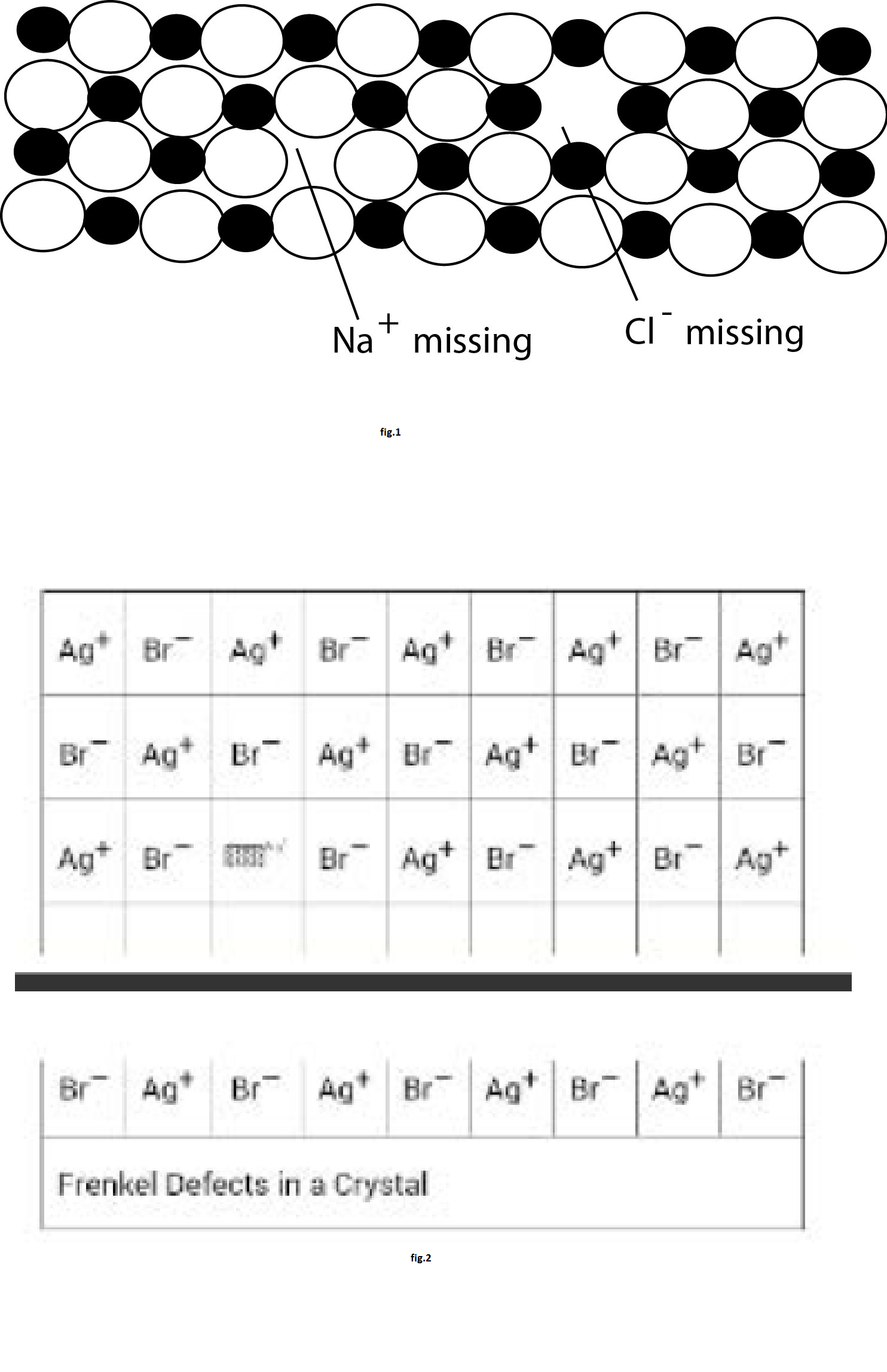
`text(Schottky Defects)` : A vacancy at a cation site is mostly accompanied by a vacancy at a nearby anion site. Such paired cation-anion vacancies are referred as Schottky defect. Such defect preserves the electrical neutrality of the crystal but the density of crystal decreases. The points which are unoccupied are called lattice vacancies. In `NaCl` the existence of two vacancies, one due to a missing `Na^(+)` ion and the other due to a missing `Cl^-` ion in a crystal of `NaCl`, is shown in fig.1. The crystal, as a whole remains neutral because the number of missing positive and negative ions is the same. Thus a Schottky defects consists of a pair of holes in the crystal lattice.
`text(Frenkel Defects)` : When an ion (cation or anion) leaves its lattice point and occupies some interstitial space, the defect is called Frenkel defect. This defect also preserves the electrical neutrality of the crystal and the density of the crystal also remains unaltered as shown in fig.2 for the crystal of `AgBr`. As can be seen, one of the `Ag^+` ions occupies a position in the interstitial space rather than its own appropriate site in the lattice. A vacancy is thus created in the lattice as shown. It may be noted again that the crystal remains neutral since the number of positive ions is the same as the number of negative ions. The presence of `Ag^+` ions in the interstitial space of `AgBr` crystal is responsible for the formation of a photographic image on exposure of `AgBr` crystals (i.e., photographic plate) to light. `ZnS` is another crystal in which Frenkel defects appear. `Zn^(2+)` ions are entrapped in the interstitial space leaving vacancies in the lattice. Frenkel defects appear in crystals in which the negative ions are much larger than the positive ions. Like Schottky defects, the Frenkel defects are also responsible for the conduction of electricity in crystals and also for the phenomenon of diffusion in solids.
`text(Schottky Defects)` : A vacancy at a cation site is mostly accompanied by a vacancy at a nearby anion site. Such paired cation-anion vacancies are referred as Schottky defect. Such defect preserves the electrical neutrality of the crystal but the density of crystal decreases. The points which are unoccupied are called lattice vacancies. In `NaCl` the existence of two vacancies, one due to a missing `Na^(+)` ion and the other due to a missing `Cl^-` ion in a crystal of `NaCl`, is shown in fig.1. The crystal, as a whole remains neutral because the number of missing positive and negative ions is the same. Thus a Schottky defects consists of a pair of holes in the crystal lattice.
`text(Frenkel Defects)` : When an ion (cation or anion) leaves its lattice point and occupies some interstitial space, the defect is called Frenkel defect. This defect also preserves the electrical neutrality of the crystal and the density of the crystal also remains unaltered as shown in fig.2 for the crystal of `AgBr`. As can be seen, one of the `Ag^+` ions occupies a position in the interstitial space rather than its own appropriate site in the lattice. A vacancy is thus created in the lattice as shown. It may be noted again that the crystal remains neutral since the number of positive ions is the same as the number of negative ions. The presence of `Ag^+` ions in the interstitial space of `AgBr` crystal is responsible for the formation of a photographic image on exposure of `AgBr` crystals (i.e., photographic plate) to light. `ZnS` is another crystal in which Frenkel defects appear. `Zn^(2+)` ions are entrapped in the interstitial space leaving vacancies in the lattice. Frenkel defects appear in crystals in which the negative ions are much larger than the positive ions. Like Schottky defects, the Frenkel defects are also responsible for the conduction of electricity in crystals and also for the phenomenon of diffusion in solids.