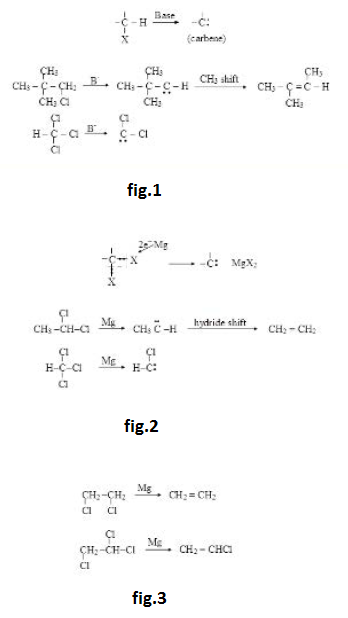
Leaving Group & Leaving Tendency :
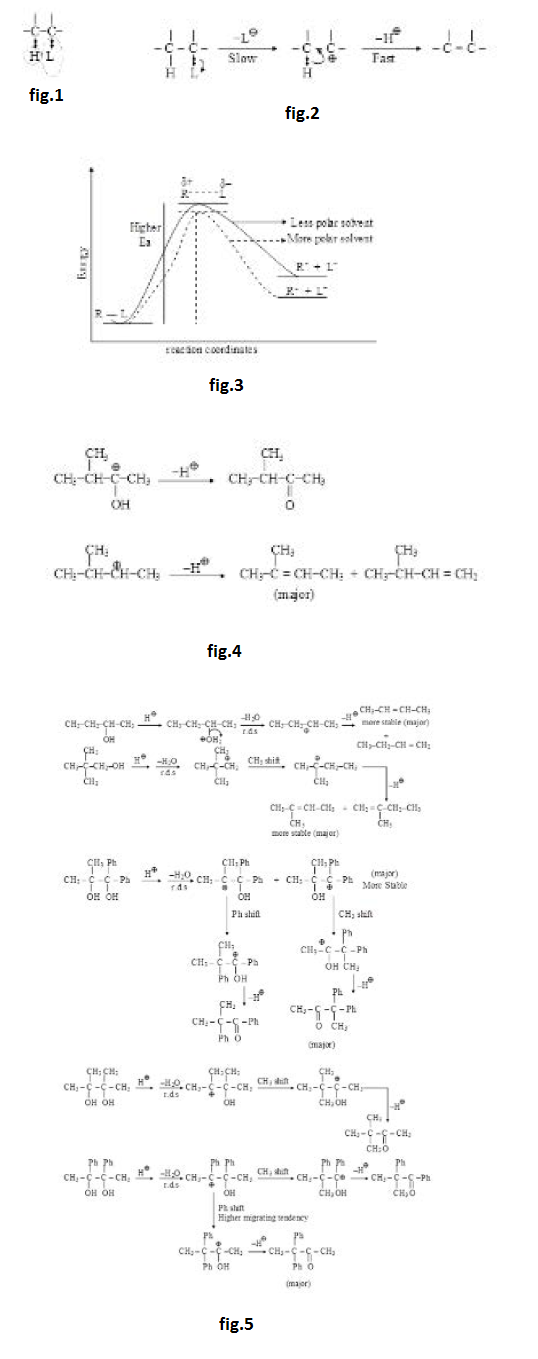
See fig.1.
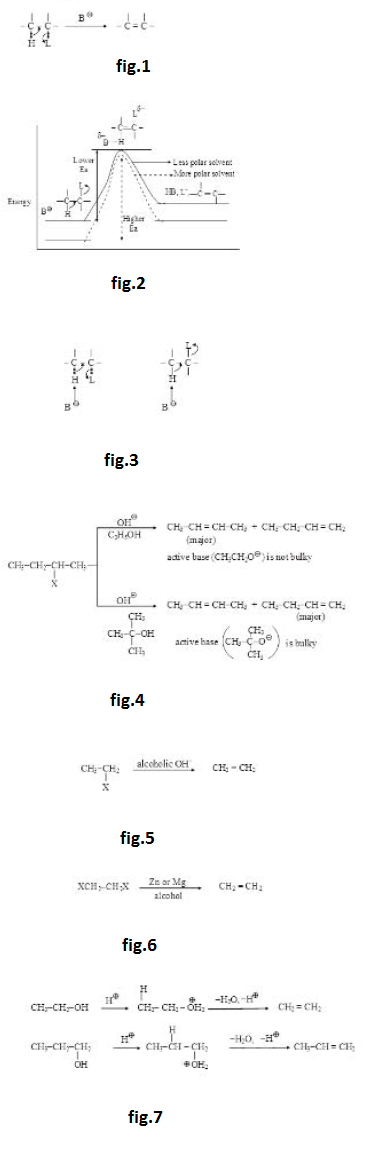
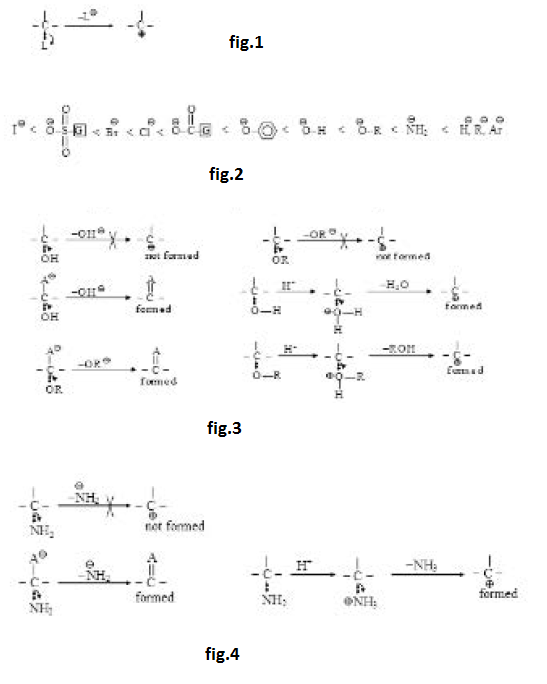
The group which is removed with the bonding electron is called leaving group and is represented by `'L'`. The ease of removal of `L` (leaving tendency) is inversely related to the basic strength of `L`. Weaker bases are better leaving groups whereas stronger bases are poorer leaving groups. The basic strength of few leaving groups increases as below : See fig.2
(i) On the basis of basic strength we can say that halides are very good leaving groups and their leaving tendency is `I^(-) > Br^(-) > Cl^(-)` as their basic strength is `I^(-) < Br^(-) < Cl^(-) < F^(-)`. `F^(-)` is considered as bad leaving group among halides.
(ii) `RCOO^(-)` is weak base and hence a good leaving group. This group can be removed easily.
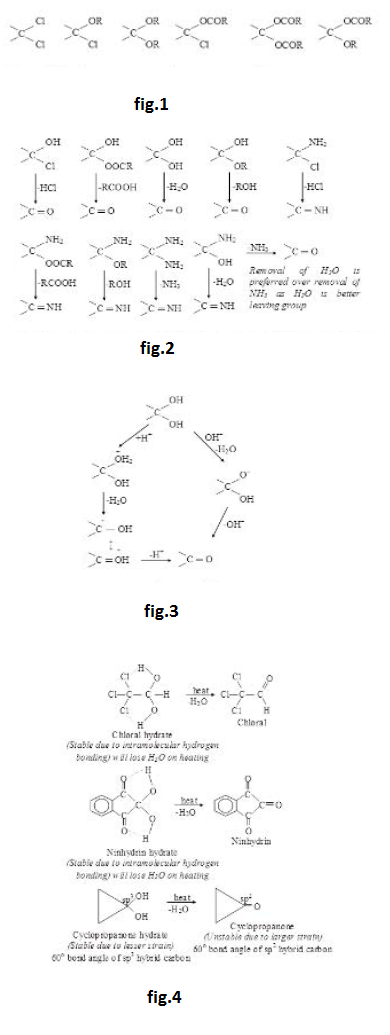
(iii) `HO^(-)` and `RO^(-)` are strong bases and hence bad leaving groups as such. They are not generally removed as such from neutral molecules but can be removed from anions. From neutral molecules, they can be removed very easily after their protonation in the form of `H_2O` and `ROH` which are very weak bases (almost neutral). See fig.3.
(iv) Removal of `text( )^(-)NH_2` is very difficult as it is a very strong base. It can not be removed as such from neutral molecules but can be removed (not so easily) from anions. It may also be removed (not so easily) from neutral molecules after its protonation in the form of ammonia which is a weak base. See fig.4.
(v) Removal of `H^(-), R^(-)` and `Ar^(-)` is very-very difficult as these are very-very strong bases and they can not be even protonated. Hence `H^(-), R^(-)` and `Ar^(-)` are not generally considered as leaving groups. Their removal is almost impossible (almost a last option) even from anion. Moreover, if at all they are removed from anions, their further reaction takes place in same step.
The group which is removed with the bonding electron is called leaving group and is represented by `'L'`. The ease of removal of `L` (leaving tendency) is inversely related to the basic strength of `L`. Weaker bases are better leaving groups whereas stronger bases are poorer leaving groups. The basic strength of few leaving groups increases as below : See fig.2
(i) On the basis of basic strength we can say that halides are very good leaving groups and their leaving tendency is `I^(-) > Br^(-) > Cl^(-)` as their basic strength is `I^(-) < Br^(-) < Cl^(-) < F^(-)`. `F^(-)` is considered as bad leaving group among halides.
(ii) `RCOO^(-)` is weak base and hence a good leaving group. This group can be removed easily.
(iii) `HO^(-)` and `RO^(-)` are strong bases and hence bad leaving groups as such. They are not generally removed as such from neutral molecules but can be removed from anions. From neutral molecules, they can be removed very easily after their protonation in the form of `H_2O` and `ROH` which are very weak bases (almost neutral). See fig.3.
(iv) Removal of `text( )^(-)NH_2` is very difficult as it is a very strong base. It can not be removed as such from neutral molecules but can be removed (not so easily) from anions. It may also be removed (not so easily) from neutral molecules after its protonation in the form of ammonia which is a weak base. See fig.4.
(v) Removal of `H^(-), R^(-)` and `Ar^(-)` is very-very difficult as these are very-very strong bases and they can not be even protonated. Hence `H^(-), R^(-)` and `Ar^(-)` are not generally considered as leaving groups. Their removal is almost impossible (almost a last option) even from anion. Moreover, if at all they are removed from anions, their further reaction takes place in same step.