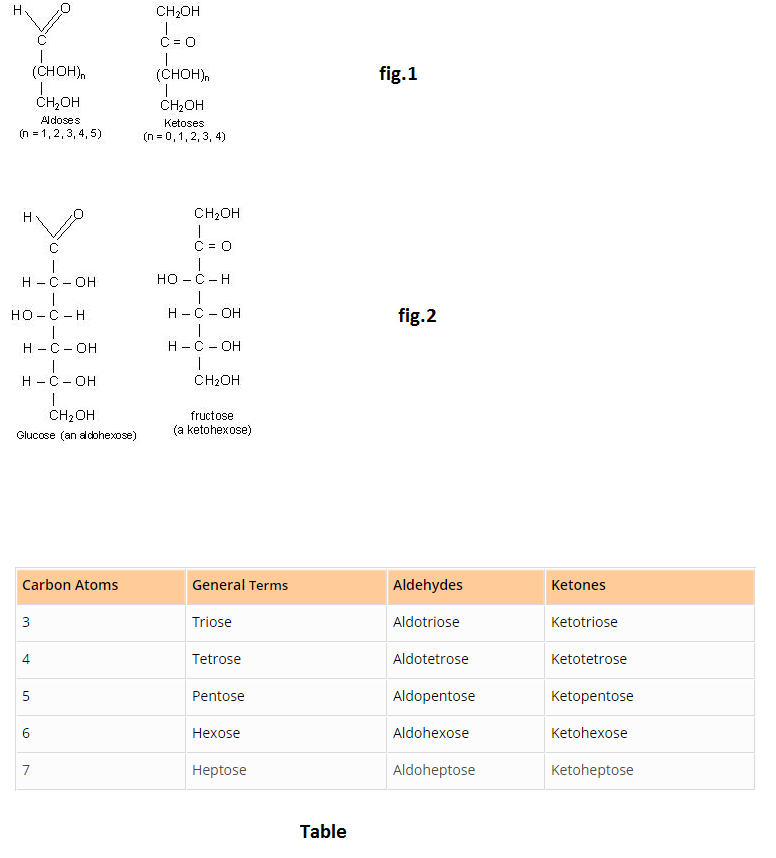
The polysaccharides are carbohydrates of high molecular weight which yield many monosaccharide molecules on hydrolysis. Examples are starch and cellulose, both of which have molecular formula, `(C_6H_(10)O_5)_n` .
`undersettext(starch)((C_6H_(10)O_5)_n) +nH_2O overset(H^(+))-> undersettext(glucose)(nC_6H_(12)O_6)`
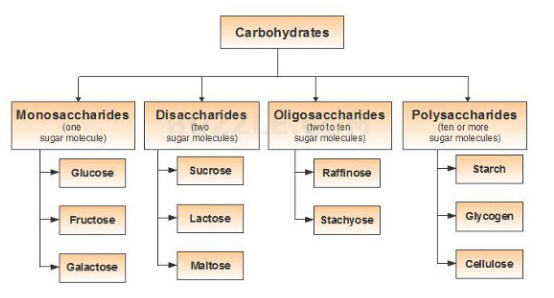
In general, the monosaccharides and oligosaccharides are crystalline solids, soluble in water and sweet to taste. They are collectively known as sugars. The polysaccharides, on the other hand, are amorphous, insoluble in water and tasteless. They are called non-sugars. The carbohydrates may also be classified as either reducing or non-reducing sugars. All those carbohydrates which have the ability to reduce Fehling’s solution and Tollen’s reagent are referred to as reducing sugars, while others are non-reducing sugars. All monosaccharides and the disaccharides other than sucrose are reducing sugars.Polysaccharides are formed when a large number (hundreds to even thousands) of monosaccharide molecules join together with the elimination of water molecule. Thus, polysaccharides may be regarded as condensation polymers in which the monosaccharides are joined together by glycosidic linkages.
Some important polysaccharides are:
1. Cellulose
2. Starch
3. Glycogen
4. Gums and
5. Pectins
6. Starch
It is a polymer of glucose. Its molecular formula is (C6H10O5)n where the value of n (200 – 1000) varies from source to source. It is the chief food reserve material or storage polysaccharide of plants and is found mainly in seeds, roots, tubers, etc. Wheat, rice, potatoes, corn, bananas etc., are rich sources of starch.
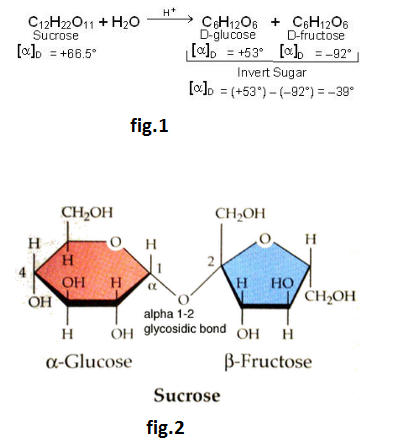
Starch is not a single compound but is a mixture of two components – amylose (10 to 20%) and amylopectin (20 to 80%). Both amylose and amylopectin are polymers of α-D-glucose.
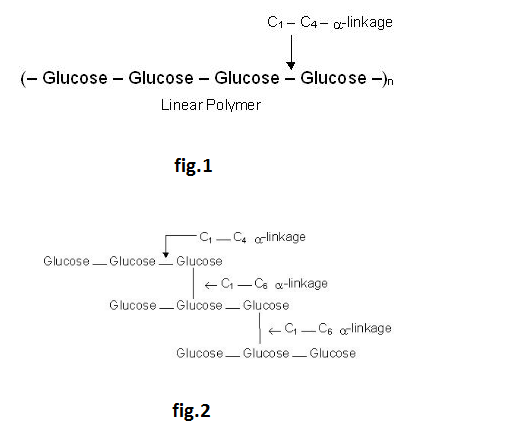
Amylose is a linear polymer of α-D-glucose. It contains about 200 glucose units which are linked to one another through α-linkage involving C1 of one glucose unit with C4 of the other as shown below: See fig.1.
Amylopectin, on the other hand, is a highly branched polymer. It consists of a large number (several branches) of short chains each containing 20-25 glucose units which are joined together through α-linkages involving C1 of one glucose unit with C4of the other. The C1 of terminal glucose unit in each chain is further linked to C6 of the other glucose unit in the next chain through C1 – C6 α-linkage. This gives amylopectin a highly branched structure as shown below- See fig.2.
Hydrolysis: Hydrolysis of starch with hot dilute acids or by enzymes gives dextrins of varying complexity, maltose and finally D-glucose. Starch does not reduce Tollen’s reagent and Fehling’s solution.
Uses: It is used as a food. It is encountered daily in the form of potatoes, bread, cakes, rice etc. It is used in coating and sizing paper to improve the writing qualities. Starch is used to treat textile fibres before they are woven into cloth so that they can be woven without breaking. It is used in manufacture of dextrins, glucose and ethyl alcohol. Starch is also used in manufacture of starch nitrate, which is used as an explosive.
The polysaccharides are carbohydrates of high molecular weight which yield many monosaccharide molecules on hydrolysis. Examples are starch and cellulose, both of which have molecular formula, `(C_6H_(10)O_5)_n` .
`undersettext(starch)((C_6H_(10)O_5)_n) +nH_2O overset(H^(+))-> undersettext(glucose)(nC_6H_(12)O_6)`
In general, the monosaccharides and oligosaccharides are crystalline solids, soluble in water and sweet to taste. They are collectively known as sugars. The polysaccharides, on the other hand, are amorphous, insoluble in water and tasteless. They are called non-sugars. The carbohydrates may also be classified as either reducing or non-reducing sugars. All those carbohydrates which have the ability to reduce Fehling’s solution and Tollen’s reagent are referred to as reducing sugars, while others are non-reducing sugars. All monosaccharides and the disaccharides other than sucrose are reducing sugars.Polysaccharides are formed when a large number (hundreds to even thousands) of monosaccharide molecules join together with the elimination of water molecule. Thus, polysaccharides may be regarded as condensation polymers in which the monosaccharides are joined together by glycosidic linkages.
Some important polysaccharides are:
1. Cellulose
2. Starch
3. Glycogen
4. Gums and
5. Pectins
6. Starch
It is a polymer of glucose. Its molecular formula is (C6H10O5)n where the value of n (200 – 1000) varies from source to source. It is the chief food reserve material or storage polysaccharide of plants and is found mainly in seeds, roots, tubers, etc. Wheat, rice, potatoes, corn, bananas etc., are rich sources of starch.
Starch is not a single compound but is a mixture of two components – amylose (10 to 20%) and amylopectin (20 to 80%). Both amylose and amylopectin are polymers of α-D-glucose.
Amylose is a linear polymer of α-D-glucose. It contains about 200 glucose units which are linked to one another through α-linkage involving C1 of one glucose unit with C4 of the other as shown below: See fig.1.
Amylopectin, on the other hand, is a highly branched polymer. It consists of a large number (several branches) of short chains each containing 20-25 glucose units which are joined together through α-linkages involving C1 of one glucose unit with C4of the other. The C1 of terminal glucose unit in each chain is further linked to C6 of the other glucose unit in the next chain through C1 – C6 α-linkage. This gives amylopectin a highly branched structure as shown below- See fig.2.
Hydrolysis: Hydrolysis of starch with hot dilute acids or by enzymes gives dextrins of varying complexity, maltose and finally D-glucose. Starch does not reduce Tollen’s reagent and Fehling’s solution.
Uses: It is used as a food. It is encountered daily in the form of potatoes, bread, cakes, rice etc. It is used in coating and sizing paper to improve the writing qualities. Starch is used to treat textile fibres before they are woven into cloth so that they can be woven without breaking. It is used in manufacture of dextrins, glucose and ethyl alcohol. Starch is also used in manufacture of starch nitrate, which is used as an explosive.