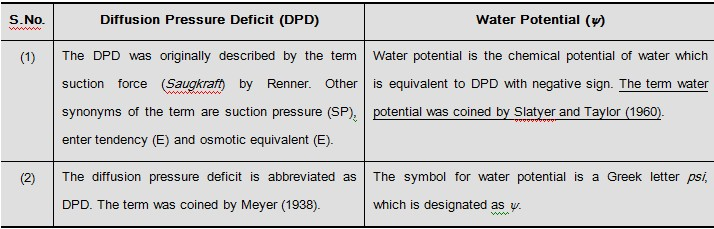
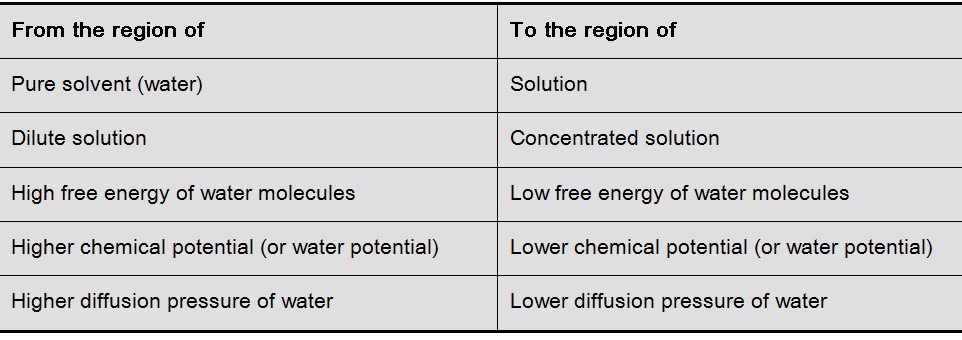
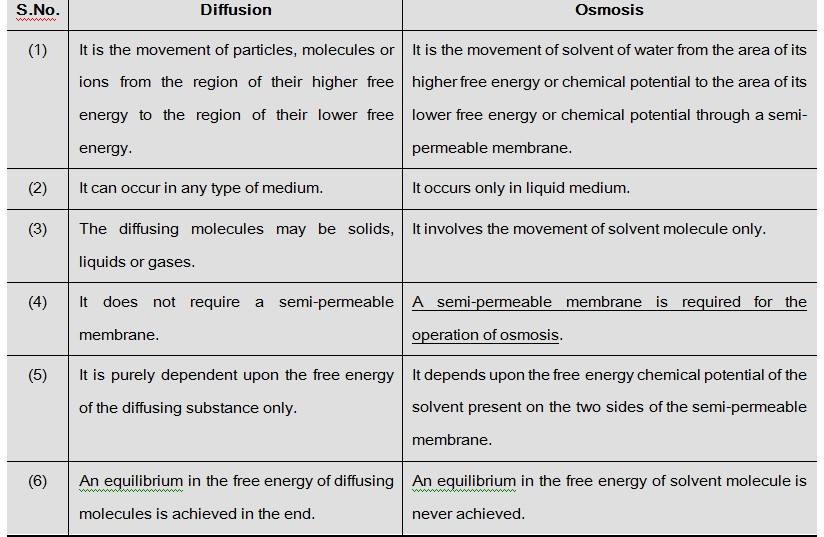
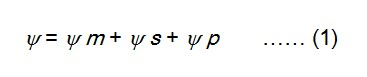A solution can be termed as hypotonic, hypertonic and isotonic depending upon its osmotic concentration, with respect to another solution or cell sap.
# (a) Hypotonic solution (hypo = less than). A solution, whose osmotic concentration (solute potential) is less than that of another solution or cell sap is called hypotonic solution. If a cell is placed in such a solution, water start moving into the cell by the process of endosmosis, and cell become turgid.
# (b) Hypertonic solution (hper = more than). A solution, whose osmotic concentration (solute potential) is more than that of another solution or cell sap is called hypertonic solution. If a cell is placed in such a solution, water comes out of the cell by the process of exosmosis and cell become flaccid. If potato tuber is placed in concentrated salt solution it would become shrink due to loss of water from its cell.
# (c) Isotonic solution (iso = the same). A solution, whose osmotic concentration (solute potential) is equal to that of another solution or cell sap, is called isotonic solution. If a cell is placed in isotonic solution, there is no net changes of water between the cell and the solution and the shape of cell remain unchanged. The normal saline (0.85% solution of NaCl) and 0.4 m to 0.5 m solution of sucrose are isotonic to the cell sap.
Osmotic concentration of a solution may governed by concentration of solute, temperature of solution, ionization of solutes and hydration of the solute molecules.
In xerophytes, the osmotic concentration of cell sap is more than normal. e.g., A molar solution of sucrose separated from pure water by such a membrane has an OP of approximately 22.4 atmospheres at 0°C. The osmotic pressure of given solution can be calculated by using the following relationship.
[ Osmotic pressure = CST ]
Where, C = Molar concentration of solution, S = Solution constant, which is 0.082 and T = Absolute temperature i.e., 273°C.
- Sucrose is non-ionizing substance while NaCl is ionizing substance. Osmotic pressure of a solution of ionizing substance is greater than that of equimolar concentration of non-ionizing substance. e.g., 0.1M sucrose solution has an OP of 2.3 bars while 0.1M sodium chloride solution has value of 4.5 bars.
A solution can be termed as hypotonic, hypertonic and isotonic depending upon its osmotic concentration, with respect to another solution or cell sap.
# (a) Hypotonic solution (hypo = less than). A solution, whose osmotic concentration (solute potential) is less than that of another solution or cell sap is called hypotonic solution. If a cell is placed in such a solution, water start moving into the cell by the process of endosmosis, and cell become turgid.
# (b) Hypertonic solution (hper = more than). A solution, whose osmotic concentration (solute potential) is more than that of another solution or cell sap is called hypertonic solution. If a cell is placed in such a solution, water comes out of the cell by the process of exosmosis and cell become flaccid. If potato tuber is placed in concentrated salt solution it would become shrink due to loss of water from its cell.
# (c) Isotonic solution (iso = the same). A solution, whose osmotic concentration (solute potential) is equal to that of another solution or cell sap, is called isotonic solution. If a cell is placed in isotonic solution, there is no net changes of water between the cell and the solution and the shape of cell remain unchanged. The normal saline (0.85% solution of NaCl) and 0.4 m to 0.5 m solution of sucrose are isotonic to the cell sap.
Osmotic concentration of a solution may governed by concentration of solute, temperature of solution, ionization of solutes and hydration of the solute molecules.
In xerophytes, the osmotic concentration of cell sap is more than normal. e.g., A molar solution of sucrose separated from pure water by such a membrane has an OP of approximately 22.4 atmospheres at 0°C. The osmotic pressure of given solution can be calculated by using the following relationship.
[ Osmotic pressure = CST ]
Where, C = Molar concentration of solution, S = Solution constant, which is 0.082 and T = Absolute temperature i.e., 273°C.
- Sucrose is non-ionizing substance while NaCl is ionizing substance. Osmotic pressure of a solution of ionizing substance is greater than that of equimolar concentration of non-ionizing substance. e.g., 0.1M sucrose solution has an OP of 2.3 bars while 0.1M sodium chloride solution has value of 4.5 bars.