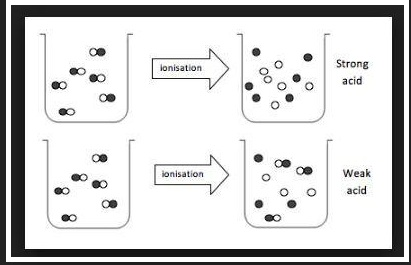
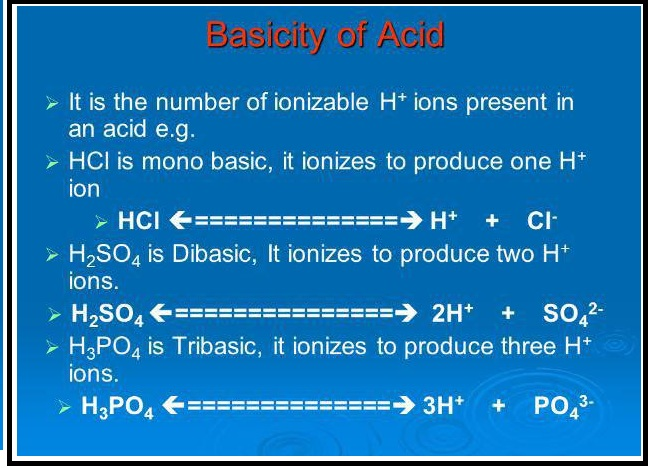
Acids have the following general properties
`=>` Acids liberates hydrogen with reactive metals like `Zn, Fe, Na`.
`Zn + 2 HCl -> ZnCl_2 + H_2 ↑`
`=>` Acids produces `CO_2` gas with carbonates and bicarbonates which turns lime water milky.
`undersettext { Marble (calcium carbonate)} ( CaCO_3 ) +undersettext ( Sulphuric acid ) (H_2SO_4) -> undersettext(Calcium suphate ) (CaSO_4) + undersettext (water) (H_2O) + undersettext (carbon dioxide) (CO_2 ↑)`
`=>` Acid reacts with base to give salt and water. It is known as neutralisation reaction.
`HCl (aq) + NaOH (aq) -> NaCl (aq) + H_2 O(l)`
`=>` Acids react with metallic oxides to form salt and water.
`Na_2 O + 2HCl -> undersettext (salt) (2 Na Cl) + H_2O`
Above reaction is similar to neutralisation reaction (Acid-base reaction). Therefore metallic oxides are also called basic oxides.
`=>` Aqueous solution of acids conduct electricity.
`=>` A concentrated solution of 3 part conc. `HCl` and 1 part conc. `HNO_3` is called aqua-regia. All metals dissolve in it.
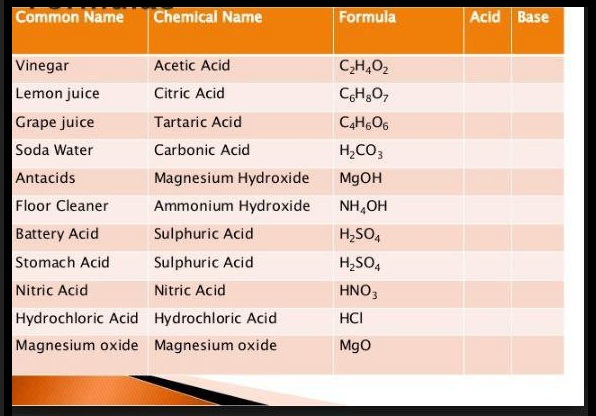
`\ \ \ \ \ \ \ \ text(Some Naturally Occurring Acids)`
| Natural source | Acids |
|---|
| Vinegar | Acetic acid |
| Orange/Lemon | Citric acid |
| Tamarind | Tartaric acid |
| Tomato | Oxalic acid |
| Curd/Milk | Lactic acid |
| Nettle sting | Methanoic acid ( formic acid) |
| Ant sting | Methanoic acid ( formic acid ) |
| Apple | Maleic acid |
| Curd | Lactic acid |
Acids have the following general properties
`=>` Acids liberates hydrogen with reactive metals like `Zn, Fe, Na`.
`Zn + 2 HCl -> ZnCl_2 + H_2 ↑`
`=>` Acids produces `CO_2` gas with carbonates and bicarbonates which turns lime water milky.
`undersettext { Marble (calcium carbonate)} ( CaCO_3 ) +undersettext ( Sulphuric acid ) (H_2SO_4) -> undersettext(Calcium suphate ) (CaSO_4) + undersettext (water) (H_2O) + undersettext (carbon dioxide) (CO_2 ↑)`
`=>` Acid reacts with base to give salt and water. It is known as neutralisation reaction.
`HCl (aq) + NaOH (aq) -> NaCl (aq) + H_2 O(l)`
`=>` Acids react with metallic oxides to form salt and water.
`Na_2 O + 2HCl -> undersettext (salt) (2 Na Cl) + H_2O`
Above reaction is similar to neutralisation reaction (Acid-base reaction). Therefore metallic oxides are also called basic oxides.
`=>` Aqueous solution of acids conduct electricity.
`=>` A concentrated solution of 3 part conc. `HCl` and 1 part conc. `HNO_3` is called aqua-regia. All metals dissolve in it.
`\ \ \ \ \ \ \ \ text(Some Naturally Occurring Acids)`
| Natural source | Acids |
|---|
| Vinegar | Acetic acid |
| Orange/Lemon | Citric acid |
| Tamarind | Tartaric acid |
| Tomato | Oxalic acid |
| Curd/Milk | Lactic acid |
| Nettle sting | Methanoic acid ( formic acid) |
| Ant sting | Methanoic acid ( formic acid ) |
| Apple | Maleic acid |
| Curd | Lactic acid |